Contact: Dan Krotz, (510) 486-4019, [email protected]
BERKELEY, CA — The first-ever glimpse of nanoscale catalysts in action could lead to improved pollution control and fuel cell technologies. Scientists from the U.S. Department of Energy’s Lawrence Berkeley National Laboratory observed catalysts restructuring themselves in response to various gases swirling around them, like a chameleon changing its color to match its surroundings.
Using a state-of-the-art spectroscopy system at Berkeley Lab’s Advanced Light Source, the team watched, for the first time, as nanoparticles composed of two catalytic metals changed their composition in the presence of different reactants. Until now, scientists have had to rely on snapshots of catalysts taken before and after a reaction, never during.
Why watch catalysts in action? Berkeley Lab's Miquel Salmeron and Gabor Somorjai discuss how a ringside seat to fundamental chemistry could lead to more efficient catalysts, cleaner skies, and less industrial waste.
This new window could give scientists the ability to develop cheaper and smarter catalysts that are fine-tuned to drive the chemistry of everyday life, such as reactions that sweep toxins from pollutants, feed hydrogen fuel cells, and drive fuel refinement techniques.
It could also expedite the development of catalysts that mop up all the substances in a reaction except the desired product, the hallmark of “green chemistry” in which waste byproducts are minimized.
“Now we can dream,” says Gabor Somorjai, a renowned surface science and catalysis expert who holds joint appointments with Berkeley Lab’s Materials Sciences Division and UC Berkeley’s department of chemistry. He conducted the research with Miquel Salmeron, a pioneer in a field of spectroscopy that enabled this work. Salmeron also holds joint appointments with Berkeley Lab’s Materials Sciences Division and UC Berkeley’s department of materials sciences and engineering.
“By watching catalysts change in real time, we can possibly design smart catalysts that optimally change as a reaction evolves,” adds Somorjai.
Catalysts, which are substances that speed up chemical reactions, are as ubiquitous as they are valuable. They’re essential to the production of many industrially important chemicals. They also play a large role in environmental chemistry, most famously exemplified by catalytic converters, which reduce toxic emissions from vehicle tailpipes.
Because of their importance, researchers all over the world are working to better understand how catalysts work, and how to improve them. Until now, however, nanoscale catalysts could only be observed before and after a reaction. The crucial segment — how a catalyst morphs during a reaction — remained the stuff of guesswork.
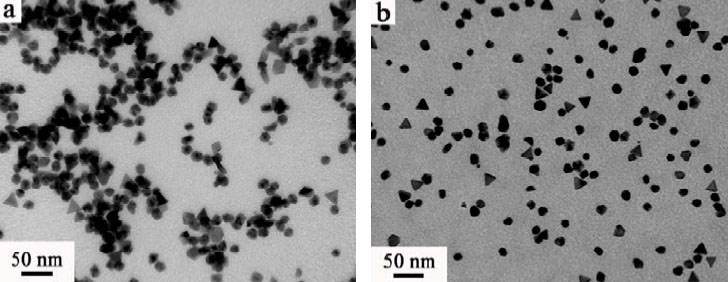
Like tiny chameleons, these rhodium-palladium nanoparticles (left) and platinum-palladium nanoparticles (right), as revealed by transmission electron microscopy, morph as their surroundings change. Tapping into this change could help streamline and guide catalytic reactions.
And that’s a huge obstacle. As Somorjai explains, it’s like trying to understand someone’s life by observing the person as a newborn baby, then fast-forwarding to old age. What transpires in between is incredibly important, but also incredibly difficult to decipher by observing two widely disparate stages.
“It’s difficult to tune a catalyst to do exactly what you want unless you know how it adapts during a reaction,” adds Salmeron. “With our work, we can for the first time see what the catalyst is doing during the reaction, not before and after.”
This new vantage required the combined expertise of both Somorjai and Salmeron. First, they had to work at the nanoscale, which is the scale at which most catalysts operate (one nanometer is one-billionth of a meter). Using techniques developed in his lab, Somorjai synthesized nanoscale particles composed of common catalytic metals. Some particles were made of rhodium and palladium, while others were made of platinum and palladium.
Next, to see how these bimetallic catalysts change in the presence of reactants, they turned to one of the few spectroscopy instruments in the world that enables scientists to study catalytic and biological phenomena in their natural environment, that is, at almost normal pressures and in the presence of different chemicals.
The instrument, the first of its kind, was developed by Salmeron and colleagues and is located at the Advanced Light Source. Like all spectroscopy systems, it identifies elements by detecting their unique spectral signals. But unlike most, the ambient pressure photoelectron spectroscopy system works under similar pressures and environments faced by everyday phenomena, instead of requiring a carefully controlled vacuum.
Using this system, the scientists watched, in real time, as the bimetallic nanoparticles restructured themselves when exposed to different gases, such as nitrogen oxide, carbon monoxide, and hydrogen. In the presence of some reactants, rhodium rose to a particle’s surface. While in the presence of other reactants, palladium rose to the surface.
“
Smarter is the trend. The more we learn about how a catalyst adapts during a reaction, the better we can design efficient catalytic reactions — Miquel Salmeron
“From one gas to another, we observed different metals segregating to a catalyst’s surface, which is the part of the catalyst that drives chemical reactions,” says Somorjai. “And this makes all the difference in establishing how the catalyst participates in the chemistry.”
With this information, scientists can develop nanoparticle catalysts and reactants that are tailored to most efficiently yield a product, whether it’s gasoline or cleaner emissions. For example, researchers can engineer bimetallic nanoparticle catalysts in which one metal rises to the surface during an initial stage of a reaction, and a different metal rises to the surface in a latter stage. The goal is to ensure that the most active metal is on the catalyst’s surface precisely when it’s needed most. In this way, the final product can be developed as quickly and cheaply as possible.
Somorjai and Salmeron hope to next observe how catalysts change shape during a reaction, which could be as equally important as compositional change in driving chemical reactions.
“Smarter is the trend,” says Salmeron. “The more we learn about how a catalyst adapts during a reaction, the better we can design efficient catalytic reactions.”
“Reaction-Driven Restructuring of Rh-Pd and Pt-Pd Core-Shell Nanoparticles” was published online Oct. 9 in Science Express. This work was supported by the Department of Energy.
Berkeley Lab is a U.S. Department of Energy national laboratory located in Berkeley, California. It conducts unclassified scientific research and is managed by the University of California. Visit our website at http://www.lbl.gov.
Additional Information
- Learn more about Gabor Somorjai’s contribution to chemistry.
- Discover how Miquel Salmeron’s explores atomic scale properties of surfaces.
- This research was conducted at the Advanced Light Source’s beamline 9.3.2.