
Like ordinary hydrogen, a single electron orbiting a proton nucleus, antihydrogen is the simplest of atoms, a single positron (antielectron) orbiting a single antiproton. CERN’s ALPHA experiment was first to trap antihydrogen in a magnetic bottle, using a superconducting octupole magnet. (Images by Chukman So, copyright © 2011 Wurtele Research Group. All rights reserved.)
The ALPHA collaboration at CERN in Geneva has scored another coup on the antimatter front by performing the first-ever spectroscopic measurements of the internal state of the antihydrogen atom. Their results are reported in a forthcoming issue of Nature and are now online.
Ordinary hydrogen atoms are the most plentiful in the universe, and also the simplest – so simple, in fact, that some of the most fundamental physical constants have been discovered by measuring the tiny energy shifts resulting from the magnetic and electric interactions of hydrogen’s proton nucleus with its single orbiting electron.
Antihydrogen, on the other hand, is rare, with single positrons (antielectrons) orbiting single antiprotons – difficult to make, and even more difficult to hold onto. Indeed antihydrogen had never been trapped until ALPHA succeeded in doing so in 2010.
In a recent series of trials, the ALPHA researchers created and captured hundreds of antihydrogen atoms in a magnetic bottle, then probed their internal states by bathing them in microwave radiation that flipped the spins of the positrons, causing the immediate ejection of the atoms from the magnetic trap and their annihilation on the trap wall.
Neither electrons nor positrons really spin, of course. “Spin” is the name for an internal quantum state of some particles and has just two values, up and down. In hydrogen, the interaction of the spin states of the electron and proton splits the ground state (the atom’s lowest energy) and is known as hyperfine splitting; in astronomy, hyperfine splitting is the source of the signature 21-centimeter emission line of hydrogen.
Antihydrogen should behave the same way, and the frequency of the microwave radiation required to flip its spins thus provides a direct measure of the difference in energy between the two hyperfine states of antihydrogen.
“To measure the hyperfine structure of antihydrogen we tune the frequency of the microwaves,” says Jonathan Wurtele, a member of the Accelerator and Fusion Research Division (AFRD) at the U.S. Department of Energy’s Lawrence Berkeley National Laboratory (Berkeley Lab), a professor of physics at the University of California at Berkeley, and a long-time member of the ALPHA collaboration. As of now the measure is imprecise, because the size of the energy difference depends on the magnitude of the magnetic field in ALPHA’s antihydrogen trap, but Wurtele says, “Our newest experimental apparatus is already under construction, and these initial experiments indicate that we’ll soon have the techniques to make precise measurements.”
How to catch an anti-atom
Berkeley Lab scientists played key roles in designing and modeling ALPHA’s Minimum Magnetic Field Trap, a magnetic bottle created by superconducting magnets whose fields capture and hold antihydrogen atoms. Although electrically neutral, the separation of the anti-atoms’ negatively charged antiprotons and positively charged positrons, plus their spins, gives them a magnetic moment. Thus – as long as they aren’t moving too fast – they are vulnerable to capture by the trap’s magnetic fields.
One advantage of working with antimatter is that it is easy to work with individual atoms, which is not the case for ordinary hydrogen. Joel Fajans, a founding member of ALPHA who is also a member of AFRD and a physics professor at UC Berkeley, explains that if one were trying to capture ordinary hydrogen atoms in a similar trap, “the vacuum in these traps is always contaminated with hydrogen, so how could you distinguish the background hydrogen from the deliberately trapped hydrogen?”
But, says Fajans, “you can’t trap antihydrogen accidentally; it just doesn’t occur naturally.” Antihydrogen, like all forms of antimatter, can’t co-exist with normal matter because matter and antimatter mutually annihilate in a burst of energy when they come in contact.
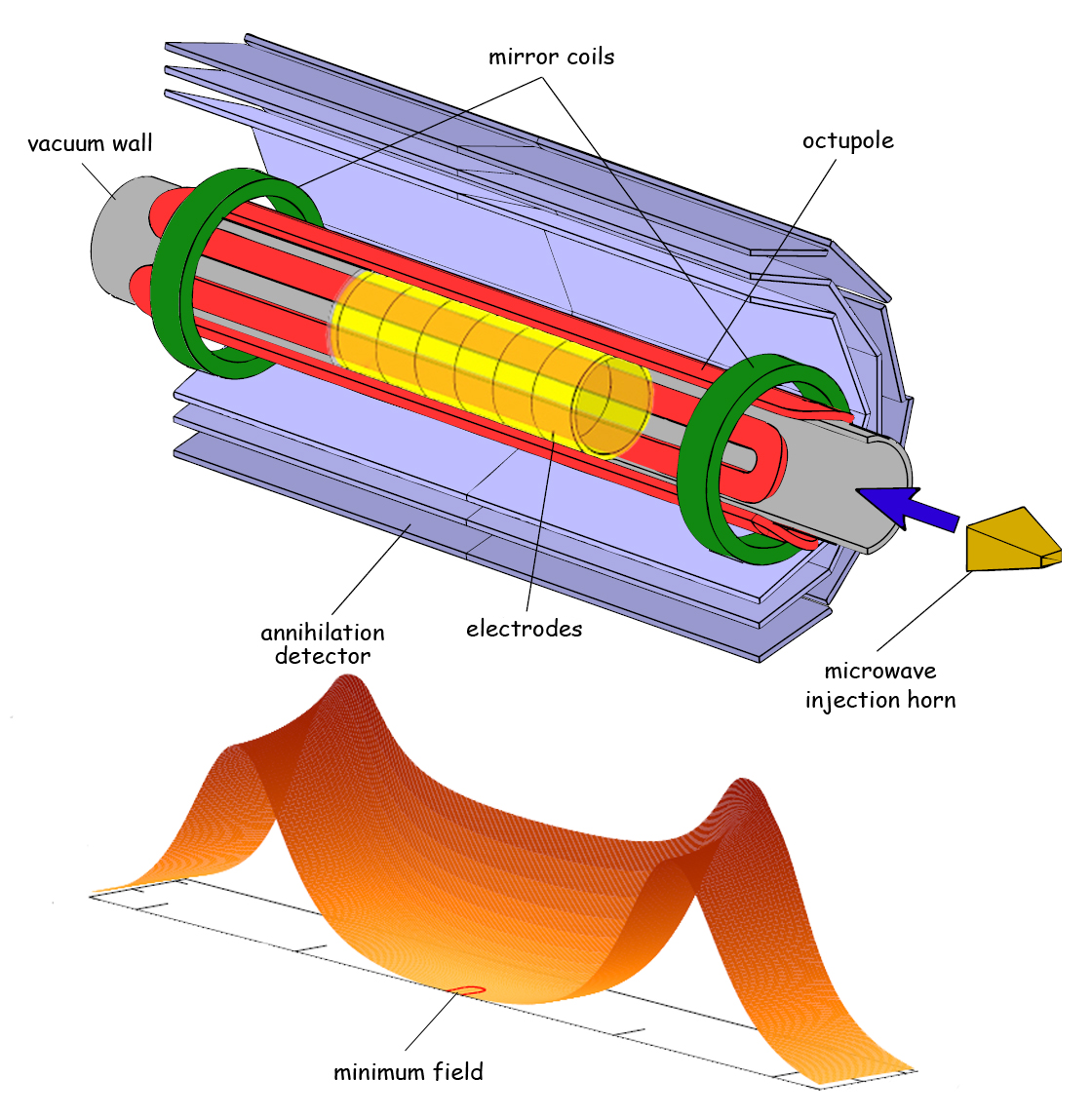
At top, a cutaway schematic of the ALPHA antimatter trap shows the superconducting octupole magnet and mirror magnets and other features. Below is a map of magnetic field strength inside the trap. The goal is to measure the hyperfine structure of antihydrogen atoms at the trap’s center, where the magnetic fields are at minimum strength. (Click on image for best resolution.)
The disadvantages of experiments on antihydrogen atoms include trying to insert experimental probes without disturbing the trap’s exquisitely balanced magnetic fields. Over the past months the ALPHA researchers have been able to modify the experiment to introduce microwave radiation into the trap’s interior. In their experiments they used two different methods of gathering data, one dubbed the disappearance method and one the appearance method.
“Disappearance” depends on establishing an average number of anti-atoms trapped in a session by abruptly turning off the trap’s superconducting magnets and counting the anti-atoms as they annihilate themselves against the experiment’s ordinary-matter walls, during the 30 thousandths of a second it takes the magnets to turn off. In some of these trials the microwaves are turned on at the so-called resonant frequency, the frequency calculated to flip the positron spins; during other trials the microwaves are on but not at the resonant frequency; and in still other trials the microwaves are not turned on at all.
Comparing the results shows that far fewer anti-atoms survive the resonant-frequency tests than in either of the other two conditions, confirming that this is indeed the right frequency to flip their spins and eject the atoms.
The “appearance” test is more direct – the anti-atoms are held in the trap for three minutes, and researchers count each annihilation that results when the microwave flips a spin and the anti-atom escapes the trap.
“One problem is that sometimes antihydrogen annihilations can be mimicked by cosmic rays,” says Fajans. “One of the postdocs in the collaboration, Simone Straka, worked on our counting algorithm, sacrificing some ability to detect all the annihilations – it has to look like a perfect antihydrogen annihilation or it won’t be counted – but gaining a factor of 10 in knowing that we weren’t being fooled by a cosmic ray. We could then turn on the microwaves and flip the spins and watch the annihilations continually.”
Large uncertainties remain. Ideally the affected anti-atoms are in the very center of the trap, where the magnetic field is at a minimum. But the field increases in every direction away from the minimum, and the magnet currents are not perfectly known. Field strength affects the resonant frequency, so the researchers have to find ways to measure the precise and accurate strength of the field where each anti-atom is flipped.
Other approaches include measuring energy shifts in the antihydrogen spectrum that are far subtler and more difficult to detect but not affected by magnetic fields. Fajans and Wurtele also look forward to a modification of ALPHA that will allow the trapped anti-atoms to be probed not by microwaves but with a laser beam, which, says Fajans, ”will allow us to measure the energy levels of antihydrogen – effectively we’ll see at what color antihydrogen glows.” Lasers also bring the ability to manipulate individual atoms in special ways, such as cooling them so that they become almost motionless, which permits ever more precise measurements of their properties.
Are matter and antimatter really the same?
“We have every reason to believe that hydrogen and antihydrogen are identical – aside from one being made of matter and the other of antimatter,” Fajans says. “In fact, CPT insists they are identical. But what if they aren’t?”
CPT stands for charge conjugation (C), parity (P), and time reversal (T), a symmetrical state of affairs in which the laws of physics are the same in a world where charges have their signs flipped, right and left are reversed, and time runs backwards.
Violations of charge conjugation and parity have been observed separately, but when the reversible arrow of time is considered, the symmetry of the three together seems to hold. The CPT mirror world would be far from a perfect reflection of ordinary reality, however – perhaps more like a funhouse hall of mirrors – if the atomic spectrum of antihydrogen is not identical to that of ordinary hydrogen.
Should antihydrogen’s spectrum turn out to be different than ordinary hydrogen, the implications range from the miniscule to the cosmic. “If we do find a difference,” says Wurtele, “people will be shocked. But we’ll never know unless we make the measurement.”
The mystery of why there is so much ordinary matter in the universe and so little antimatter has long exercised theorists and experimentalists alike. It’s at least conceivable that part of the answer lies in the spectrum of antihydrogen. If so, the ALPHA collaboration is on the path to finding it.
###
“Resonant quantum transitions in trapped antihydrogen atoms,” by C. Amole, M. D. Ashkezari, M. Baquero-Ruiz, W. Bertsche, P. D. Bowe, E. Butler, A. Capra, C. L. Cesar, M. Charlton, A. Deller, P. H. Donnan, S. Eriksson, J. Fajans, T. Friesen, M. C. Fujiwara, D. R. Gill, A. Gutierrez, J. S. Hangst, W. N. Hardy, M. E. Hayden, A. J. Humphries, C. A. Isaac, S. Jonsell, L. Kurchaninov, A. Little, N. Madsen, J. T. K. McKenna, S. Menary, S. C. Napoli, P. Nolan, K. Olchanski, A. Olin, P. Pusa, C. Ø. Rasmussen, F. Robicheaux, E. Sarid, C. R. Shields, D. M. Silveira, S. Stracka, C. So, R. I. Thompson, D. P. van der Werf, and J. S. Wurtele, appears in Nature and is available on line at http://www.nature.com/nature/journal/vaop/ncurrent/full/nature10942.html. In addition to Fajans and Wurtele, Berkeley participants in this work include UC Berkeley graduate students Marcelo Baquero-Luiz and Chukman So, and undergraduate Arielle Little.
Read the CERN press release at http://press.web.cern.ch/press/PressReleases/Releases2012/PR06.12E.html.
For more about ALPHA, visit these sites of interest:
ALPHA’s website at http://alpha-new.web.cern.ch/
The Fajans Group website at http://socrates.berkeley.edu/~fajans/
The Wurtele Group website at http://raman.physics.berkeley.edu/; especially see graphics at http://raman.physics.berkeley.edu/gallery.html
“ALPHA stores antimatter atoms over a quarter of an hour,” at http://newscenter.lbl.gov/news-releases/2011/06/05/alpha-quarter-hour/
“Antimatter atoms successfully stored for the first time,” at http://newscenter.lbl.gov/news-releases/2010/11/17/antimatter-atoms/
“Angels, demons, and antihydrogen,” at http://newscenter.lbl.gov/feature-stories/2009/05/05/angels-demons-and-antihydrogen/
U.S. participation in the international ALPHA collaboration is supported by the National Science Foundation and the U.S. Department of Energy’s Office of Science.
DOE’s Office of Science is the single largest supporter of basic research in the physical sciences in the United States, and is working to address some of the most pressing challenges of our time. For more information, please visit science.energy.gov.
Lawrence Berkeley National Laboratory addresses the world’s most urgent scientific challenges by advancing sustainable energy, protecting human health, creating new materials, and revealing the origin and fate of the universe. Founded in 1931, Berkeley Lab’s scientific expertise has been recognized with 13 Nobel prizes. The University of California manages Berkeley Lab for the U.S. Department of Energy’s Office of Science. For more, visit www.lbl.gov.