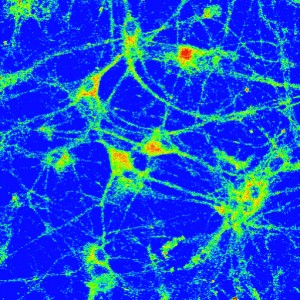
Copper Sensor-3 (CS3), a small-molecule fluorescent probe, was used to map the movement of copper in the brain triggered by neuronal activity.
The value of copper has risen dramatically in the 21st century as many a thief can tell you, but in addition to the thermal and electrical properties that make it such a hot commodity metal, copper has chemical properties that make it essential to a healthy brain. Working at the interface of chemistry and neuroscience, Berkeley Lab chemist Christopher Chang and his research group at UC Berkeley have developed a series of fluorescent probes for molecular imaging of copper in the brain. Speaking at the recent national meeting of the American Chemical Society in New Orleans, he described the challenges of creating and applying live-cell and live-animal copper imaging probes and explained the importance of meeting these challenges.
“The human brain is a unique biological system, possessing unparalleled biological complexity in a compact space,” Chang said. “Although it accounts for only two-percent of total body mass, it consumes 20-percent of the oxygen taken in through respiration. As a consequence of its high demand for oxygen and oxidative metabolism, the brain has among the highest levels of copper, as well as iron and zinc in the body.”
Neuron and glia cells in the brain both require copper for the basic respiratory and antioxidant enzymes cytochrome c oxidase and superoxide dismutase. Copper is also necessary for brain-specific enzymes that control neurotransmitters, such as dopamine, as well as neuropeptides and dietary amines. Disruption of copper oxidation in the brain has been linked to several neurodegenerative diseases, including Alzheimer’s, Parkinson’s, Menkes’ and Wilson’s.

Christopher Chang is a chemist who holds joint appointments with Berkeley Lab and UC Berkeley and is also an investigator with the Howard Hughes Medical Institute. (Photo by Roy Kaltschmidt)
“The complex relationships between copper status and various stages of health and disease have been difficult to determine in part because of a lack of methods for monitoring dynamic changes in copper pools in whole living organisms,” Chang said. “We’ve been designing fluorescent probes that can map the movement of copper in live cells, tissue or even model organisms, such as mice and zebrafish.”
Their first success was Coppersensor-3 (CS3), a small-molecule fluorescent probe that can be used to image labile copper pools in living cells at endogenous, basal levels. They used CS3 in conjunction with synchrotron-based X-ray fluorescence microscopy (XRFM) to discover that neuronal cells move significant pools of copper upon activation and that these copper movements are dependent on calcium signaling.
“This was the first established link between mobile copper and major cell signaling pathways,” Chang said. “Being able to map transient copper movements after neuronal depolarization revealed how neural activity triggers copper mobility, and enabled us to create a model for calcium/copper crosstalk in neurons.”
The CS3 probe was followed by Mitochondrial Coppersensor-1 (Mito-CS1), a fluorescent sensor that can selectively target mitochondria and detect basal and labile copper pools in living cells. Mitochondria, the organelles that generate most of the chemical energy used by cells, are important reservoirs for copper. By allowing direct, real-time visualization of exchangeable mitochondrial copper pools, the Mito-CS1 probe enabled Chang and his colleagues to discover that cells maintain copper homeostasis in mitochondria even in situations of copper deficiency and metabolic malfunctions.
“This work illustrated the importance of regulating copper stores in mitochondria,” Chang said.
The latest copper probe from Chang’s group is Coppersensor 790 (CS790), a fluorescent sensor that features near-infrared excitation and emission capabilities, ideal for penetrating thicker biological specimens. CS790 can be used to monitor fluctuations in exchangeable copper stores under basal conditions, as well as under copper overload or deficiency conditions. Chang and his group are using CS790 to study a mouse model of Wilson’s disease, a genetic disorder characterized by an accumulation of excess copper.
“The in vivo fluorescence detection of copper provided by CS790 and our other fluorescent probes is opening up unique opportunities to explore the roles that copper plays in the healthy physiology of the brain, as well as in the development and progression copper-related diseases,” Chang said.
For more information about the research of Christopher Chang, visit his Website here