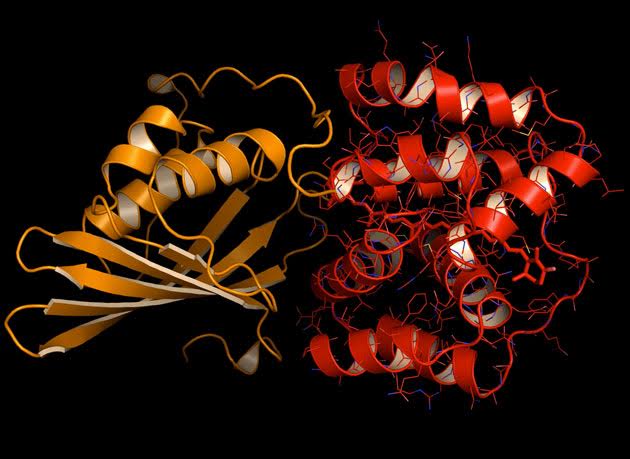
Translocation of the carotenoid pigment within a critical light-sensitive protein called the Orange Carotenoid Protein triggers a shifting of the protein from the light-absorbing orange state to the energy- quenching red state, providing cyanobacteria with protection from too much sunlight.
Overexposure to sunlight, which is damaging to natural photosynthetic systems of green plants and cyanobacteria, is also expected to be damaging to artificial photosynthetic systems. Nature has solved the problem through a photoprotection mechanism called “nonphotochemical-quenching,” in which excess solar energy is safely dissipated as heat from one molecular system to another. With an eye on learning from nature’s success, a team of Berkeley Lab researchers has discovered a surprising key event in this energy-quenching process.
In a study led by Cheryl Kerfeld, a structural biologist who holds joint appointments with Berkeley Lab’s Physical Biosciences Division and Michigan State University, the research team found that in cyanobacteria the energy-quenching mechanism is triggered by an unprecedented, large-scale movement (relatively speaking) from one location to another of the carotenoid pigment within a critical light-sensitive protein called the Orange Carotenoid Protein (OCP). As a result of this translocation, the carotenoid changes its shape slightly and interacts with a different set of amino acid neighbors causing the protein to shift from an “orange” light-absorbing state to a “red” photoprotective state. This turns out to be an unanticipated molecular priming event in photoprotection.
“Prior to our work, the assumption was that carotenoids are static, held in place by the protein scaffold,” Kerfeld says. “Having shown that the translocation of carotenoid within the protein is a functional trigger for photoprotection, scientists will need to revisit other carotenoid-binding protein complexes to see if translocation could play a role in those as well. Understanding the dynamic function of carotenoids should be useful for the design of future artificial photosynthetic systems.”

Corie Ralston and Cheryl Kerfeld at ALS Beamline 5.0.2 where crystal structures of the Orange Carotenoid Protein in cyanobacteria were recorded as the protein transitioned from light-absorber to photoprotector. (Photo by Roy Kaltschmidt)
Kerfeld is the corresponding author of a paper in Science describing this research titled “A 12 Å carotenoid translocation in a photoswitch associated with cyanobacterial photoprotection.” Co-authors are Ryan Leverenz, Markus Sutter, Adjélé Wilson, Sayan Gupta, Adrien Thurotte, Céline Bourcier de Carbon, Christopher Petzold, Corie Ralston, François Perreau and Diana Kirilovsky.
Through photosynthesis, plants are able to harvest solar energy and convert it to chemical energy. Creating an efficient artificial version of photosynthesis would realize the dream of solar power as the ultimate green and renewable source of electrical energy. However, if a sunlight-harvesting system becomes overloaded with absorbed solar energy, it most likely will suffer some form of damage.
“We know that the interactions of pigment molecules with one another and with proteins are of fundamental importance to the light harvesting and photoprotective functions essential to oxygenic photosynthesis,” Kerfeld says. “We also know that the distance between two pigment molecules is important for the transfer of energy, which means that knowing precisely a pigment molecule’s structural arrangement and its location at any given time in the process is critical.”
Kerfeld and her co-authors focused on the OCP, a pigment-binding protein in cyanobacteria that absorbs blue-green light and binds to the the cyanobacterial antenna to cause the dissipation of excess captured energy. Cyanobacteria are aquatic microbes that have been called the “architects of Earth’s atmosphere” because they generated most of the atmosphere’s oxygen during the Archaean and Proterozoic Eras. The chloroplasts that enable green plants to carry out photosynthesis today are descendants of ancient cyanobacteria.
As orange-state OCP absorbs blue-green light, it undergoes structural changes that result in red-state OCP. Once the excess solar energy has been quenched, OCP interacts with a second protein, the Fluorescence Recovery Protein (FRP), causing it to revert back to the light-harvesting orange state. Working at Berkeley Lab’s Advanced Light Source, a U.S. Department of Energy (DOE) Office of Science national user facility, Kerfeld and her co-authors used the protein crystallography capabilities of ALS Beamlines 5.0.1 and 5.0.2 to obtain crystal structures of these key photoprotective proteins.
“These crystal structures revealed that OCP photoactivation is accompanied by a 12 Å translocation of the pigment within the protein and a dramatic reconfiguration of carotenoid-protein interactions,” Kerfeld says. “Our results also identified the origin of the photochromic changes in the OCP triggered by light and revealed the structural determinants required for interaction with the light-harvesting antenna during photoprotection.”
To confirm that this translocation actually occurs when OCP is in its natural solution environment and was not due to structural changes that the protein undergoes during crystal formation, Kerfeld and her colleagues turned to ALS Beamline 5.3.1 and X-ray Footprinting (XFP), a powerful technique in structural biology for the study of macromolecular structures and dynamics of proteins and nucleic acids in solution.
“In any protein crystallography study, there is always the question about whether the crystallized protein represents the protein in real life,” says biophysicist Corie Ralston, director of the Berkeley Center for Structural Biology (BCSB), which operates the ALS Beamlines used in this study, and a co-author of the Science paper. “One of the most powerful things about XFP is that you can look at proteins and/or nucleic acids in the solution state, and often under conditions that are close to physiological.”
With the XFP study having confirmed the protein crystallography results as real, Kerfeld says the next step will be to understand the detailed mechanism behind OCP’s interaction with the antenna to dissipate energy.
“Some cyanobacteria have multiple homologs to the OCP,” she says. “What are these homologs doing? Do they allow for fine-tuning of photoprotection? If we can learn the answers we might be able to engineer smart photoprotection in cyanos or even improve photosynthesis in green plants.”
This research was supported by the DOE Office of Science.
Additional Information
For more about the research of Cheryl Kerfeld go here
For more about the Berkeley Center for Structural Biology go here
# # #
Lawrence Berkeley National Laboratory addresses the world’s most urgent scientific challenges by advancing sustainable energy, protecting human health, creating new materials, and revealing the origin and fate of the universe. Founded in 1931, Berkeley Lab’s scientific expertise has been recognized with 13 Nobel prizes. The University of California manages Berkeley Lab for the U.S. Department of Energy’s Office of Science. For more, visit www.lbl.gov.