
Metal–organic frameworks (MOFs) with flexible gas-adsorbing pores could make the driving range of adsorbed-natural-gas (ANG) cars comparable to that of a typical gasoline-powered car.
With new makes of all-electric and hybrid automobiles seeming to emerge as fast as the colors of fall, it is easy to overlook another alternative to gasoline engines that could prove to be a major player in reduced-carbon transportation – cars powered by natural gas. Natural gas, which consists primarily of methane (CH4) is an abundant, cheaper and cleaner burning fuel than gasoline, but its low energy density at ambient temperature and pressure has posed a severe challenge for on-board fuel storage in cars. Help may be on the way.
Researchers with the U.S. Department of Energy (DOE)’s Lawrence Berkeley National Laboratory (Berkeley Lab) have developed a variety of metal–organic frameworks (MOFs) – sponge-like 3D crystals with an extraordinarily large internal surface area – that feature flexible gas-adsorbing pores. This flexibility gives these MOFs a high capacity for storing methane, which in turn has the potential to help make the driving range of an adsorbed-natural-gas (ANG) car comparable to that of a typical gasoline-powered car.
“Our flexible MOFs can be used to boost the usable capacity of natural gas in a tank, reduce the heating effects associated with filling an ANG tank, and reduce the cooling effects upon discharging the gas from the ANG tank,” says Jeffrey Long, a chemist with Berkeley Lab’s Materials Sciences Division and the University of California (UC) Berkeley who is leading this research. “This ability to maximize the deliverable capacity of natural gas while also providing internal heat management during adsorption and desorption demonstrates a new concept in the storage of natural gas that provides a possible path forward for ANG applications where none was envisioned before.”

Jeff Long is chemists who holds joint appointments with Berkeley Lab and UC Berkeley. (Photo by Roy Kaltschmidt)
Long is the corresponding author of a Nature paper that describes this work entitled, “Methane storage in flexible metal–organic frameworks with intrinsic thermal management.” The lead author is Jarad Mason, a member of Long’s research group at the time of this study and now at Northwestern University. (See below for a complete list of co-authors.)
The United States holds a vast amount of proven natural gas reserves – some 360 trillion cubic feet and climbing. While compressed natural gas-fueled vehicles are already on the road, the widespread use of natural gas as a transportation fuel has been hampered by cumbersome and expensive on-board gas storage tanks and the cost of dispensing compressed natural gas to vehicles. The storage issue is especially keen for light-duty vehicles such as cars, in which the space available for on-board fuel storage is limited. ANG has the potential to store high densities of methane within a porous material at ambient temperature and moderate pressures, but designing such high-capacity systems while still managing the thermal fluctuations associated with adsorbing and desorbing the gas from the adsorbent has proven to be difficult.
The key to the success of the MOFs developed by Long, Mason and their colleagues is a “stepped” adsorption and desorption of methane gas.
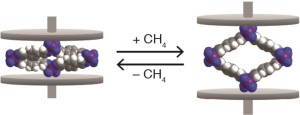
Space-filling models of cobalt-bdp MOF in the collapsed state (left) and CH4-expanded state (right). The purple, gray, blue, and white spheres represent Co, C, N, and H atoms, respectively.
“Most porous materials that would be used as adsorbents exhibit classical Langmuir-type isotherm adsorption, where the amount of methane adsorbed increases continuously but with a decreasing slope as the pressure is raised so that, upon discharging the methane down to the minimum delivery pressure, much of it remains in the tank,” Long says. “With our flexible MOFs, the adsorption process is stepped because the gas must force its way into the MOF crystal structure, opening and expanding the pores. This means the amount of methane that can be delivered to the engine, i.e., the usable capacity, is higher than for traditional, non-flexible adsorbents.”
In addition, Long says, the step in the adsorption isotherm is associated with a structural phase change in the MOF crystal that reduces the amount of heat released upon filling the tank, as well as the amount of cooling that takes place when methane is delivered to accelerate the vehicle.
“Crystallites that experience higher external pressures will have a greater free energy change associated with the phase transition and will open at higher pressures,” Long says. “Our results present the prospect of using mechanical pressure, provided, for example, through an elastic bladder, as a means of thermal management in an ANG system based on a flexible adsorbent.”
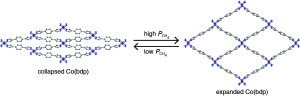
The cobalt-bdp MOF features flexible square-shaped pores that expand under pressure to adsorb increasing amounts of methane gas.
To test their approach, Long and his colleagues used a cobalt-based MOF hybrid that goes by the name “cobalt-bdp” or Co(bdp) for cobalt (benzenedipyrazolate). In its most open form, cobalt-bdp features square-shaped pores that can flex shut like an accordion when the pores are evacuated. Combined gas adsorption and in situ powder X-ray diffraction experiments performed under various pressures of methane at 25°C (77°F) showed that there is minimal adsorption of methane by the cobalt-bpd MOF at low pressures, then a sharp step upwards at 16 bar, signifying a transition from a collapsed, non-porous structure to an expanded, porous structure. This transition to an expanded phase was reversible. When the methane pressure decreased to between 10 bar and 5 bar, the framework fully converted back to the collapsed phase, pushing out all of the adsorbed methane gas.
Long says that it should be possible to design MOF adsorbents of methane with even stronger gas binding sites and higher-energy phase transitions for next generation ANG vehicles. He and his group are working on this now and are also investigating whether the strategy can be applied to hydrogen, which poses similar storage problems.
Moreover, Long says, “Improved compaction and packing strategies should also allow further reductions to external thermal-management requirements and optimization of the overall natural gas storage-system performance.”
In addition to Long and Mason, other authors of the Nature paper that describes this study were Julia Oktawiec, Mercedes Taylor, Matthew Hudson, Julien Rodriguez, Jonathan Bachman, Miguel Gonzalez, Antonio Cervellino, Antonietta Guagliardi, Craig Brown, Philip Llewellyn and Norberto Masciocchi.
This research was supported by the DOE’s Advanced Research Projects Agency – Energy (ARPA-E). The X-ray characterizations were carried out at synchrotron light sources that included the Advanced Light Source and the Advanced Photon Source, both DOE Office of Science User Facilities.
Additional Information
For more about the research of Jeffrey Long go here
# # #
Lawrence Berkeley National Laboratory addresses the world’s most urgent scientific challenges by advancing sustainable energy, protecting human health, creating new materials, and revealing the origin and fate of the universe. Founded in 1931, Berkeley Lab’s scientific expertise has been recognized with 13 Nobel prizes. The University of California manages Berkeley Lab for the U.S. Department of Energy’s Office of Science. For more, visit www.lbl.gov.
DOE’s Office of Science is the single largest supporter of basic research in the physical sciences in the United States, and is working to address some of the most pressing challenges of our time. For more information, please visit the Office of Science website at science.energy.gov/.
