Contact: Lynn Yarris (510) 486-5375, [email protected]
In revisiting a chemical reaction that’s been in the literature for several decades and adding a new wrinkle of their own, researchers with Berkeley Lab and the University of California (UC) Berkeley have discovered a mild and relatively inexpensive procedure for removing oxygen from biomass. This procedure, if it can be effectively industrialized, could allow many of today’s petrochemical products, including plastics, to instead be made from biomass.
“We’ve found and optimized a selective, one-pot deoxygenation technique based on a formic acid treatment,” said Robert Bergman, a co-principal investigator on this project who holds a joint appointment with Berkeley Lab’s Chemical Sciences Division and the UC Berkeley Chemistry Department.

(From left) Robert Bergman, Peter Marsden, Elena Arceo and Jonathan Ellman have developed a selective, high-yield, one-pot technique based on a formic acid treatment that can remove oxygen from biomass. (Photo by Roy Kaltschmidt, Berkeley Lab Public Affairs)
The formic acid, Bergman said, converts glycerol, a major and unwanted by-product in the manufacturing of biodiesel, into allyl alcohol, which is used as a starting material in the manufacturing of polymers, drugs, organic compounds, herbicides, pesticides and other chemical products. Allyl alcohol today is produced from the oxidation of petroleum.
Said Jonathan Ellman, a UC Berkeley chemistry professor and the other principal investigator in this research, “Right now, about five percent of the world’s supply of petroleum is used to make feedstocks that are synthesized into commodity chemicals. If these feedstocks can instead be made from biomass they become renewable and their production will no longer be a detriment to the environment.”
Biomass has been drawing wide public attention for its potential to be converted into carbon-neutral biofuels, but there is also huge potential for it to be converted into chemical feedstocks. It is safe to say that a day does not go by without each of us making use of multiple petrochemical products. Feedstocks for such products obtained from biomass rather than petroleum would be renewable as well as biodegradable. However, unlike petrochemical feedstocks, which are made by adding oxygen to petroleum, biomass feedstocks require the removal of oxygen from the raw material.
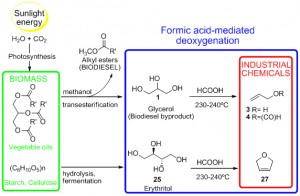
This formic acid-mediated deoxygenation reaction converts glycerol and other unwanted biomass byproducts into feedstocks for commodity chemicals. It could enable biomass to serve as a renewable replacement for petrochemicals. (graphic courtesy of Elena Arceo)
Bergman and Ellman, working with Elena Arceo, a Fulbright scholar from Spain, and Peter Marsden, a UC Berkely graduate student, used labeling experiments and a unique distillation system to take a new look at an old chemical reaction in which formic acid, the chemical found in bee venom, was used to remove oxygen from glycerol. In its original conception, the reaction was low-yielding, primarily because of substantial charring, an unselective combustion that leads to an intractable mixture under high heat. Bergman and Ellman found that simply protecting this reaction from air provided a much improved process for the deoxygenation of glycerol.
Said Bergman, “Treating glycerol with formic acid while directing a stream of nitrogen through the reaction mixture completely eliminates charring. Besides protecting the product from atmospheric oxidation, the nitrogen also facilitates distillation of the alcohol. The final product shows substantially improved yield (80-percent) and higher selectivity.”
Said Ellman, “From our studies we also gained a much better understanding of the basic chemistry behind the reaction. We thought that the charring was a random oxidation process because the reaction had been carried out in air and we expected that running it in a nitrogen environment would improve things. However, in studying the basic chemistry we uncovered an unexpected reaction pathway that really broadens the generality of this reaction and expands its potential applications.”
With this new reaction pathway, the formic acid-mediated deoxygenation technique developed by Bergman and Ellman could be used to convert the carbohydrates in biomass, as well as other polyhdroxy compounds, into the chemical feedstocks, such as olefins (alkenes) that are now derived from petroleum. The technique could also prove useful in the process by which biomass is converted into liquid transportation fuels.

Not a day goes by without our making use of multiple petrochemical products. Feedstocks for such products derived from biomass rather than petroleum would be renewable and environmentally green. (Image courtesy of Polyfort.com)
Said Bergman, “Our preliminary results with inexpensive biomass-derived polyols suggest that the reaction of polyhydroxy compounds with formic acid will be a valuable alternative for the manufacture of reduced oxygen content products. However, scaling this technique up so that biomass feedstocks are competitive with feedstocks derived from petroleum is going to be an engineering challenge.”
Added Ellman, “Scaling the technique up to industrial levels is probably going to require the combined efforts of industrial and academic laboratories, but if we are able to one day make commodity chemicals as well as fuels from biomass, we can protect the atmosphere from further damage and at the same time help lower current carbon dioxide levels.”
Berkeley Lab is a U.S. Department of Energy national laboratory located in Berkeley, California. It conducts unclassified scientific research and is managed by the University of California. Visit our Website at www.lbl.gov/
Additional Information
To learn more about the research of Robert Bergman, visit his Website at http://www.cchem.berkeley.edu/rgbgrp/index.html
To learn more about the research of Jonathan Ellman, visit his Website at http://www.cchem.berkeley.edu/jaegrp/index.html