This movie shows the conformation of Cascade’s subunits – CasA (purple), CasB (yellow) and CasE (magenta) – being rearranged while the helical spine of the complex remains relatively undisturbed.
A complex of proteins in the bacterium E.coli that plays a critical role in defending the microbe from viruses and other invaders has been discovered to have the shape of a seahorse by researchers with the U.S Department of Energy’s Lawrence Berkeley National Laboratory (Berkeley Lab). This discovery holds far more implications for your own health than you might think.
In its never-ending battle to protect you from infections by bacteria, viruses, toxins and other invasive elements, your immune system has an important ally – many allies in fact. By the time you reach adulthood, some 90-percent of the cells in your body are microbial. These microbes – collectively known as the microbiome – play a critical role in preserving the health of their human host.
“Perturbations of the human microbiome by viral and other infections can disrupt important symbioses and open the door to invasions by human pathogens,” says Blake Wiedenheft, a biochemist with Berkeley Lab and the University of California (UC) Berkeley. “By understanding the mechanisms behind microbial immune systems, we can better understand how they are similar and where they are different from the human immune system.”
Wiedenheft is part of a team of researchers, led by biochemist Jennifer Doudna, a leading authority on RNA molecular structures, and biophysicist Eva Nogales, an expert on electron microscopy and image analysis, that has provided the first sub-nanometer look at a central player in the microbial immune system. Through a combination of cryo-electron microscopy and three-dimensional image reconstruction, they have determined the structure of a protein complex called “Cascade,” that acts as a surveillance system for detecting and inactivating the nucleic acid of invading pathogens.

From left, Blake Wiedenheft, Eva Nogales, Gabriel Lander and Jennifer Doudna, used a combination of cryo-electron microscopy and 3-D image reconstruction to determine the structure of Cascade, a protein complex that detects and inactivates invading pathogens. (Photo by Roy Kaltschmidt, Berkeley Lab)
Doudna and Nogales are the corresponding authors and Wiedenheft and Gabriel Lander are the lead authors of a paper describing this research in the journal Nature. The paper is titled “Structures of the RNA-guided surveillance complex from a bacterial immune system.” Like Wiedenheft, Doudna, Nogales and Lander all hold joint appointments with Berkeley Lab and UC Berkeley. Doudna and Nogales are also investigators with the Howard Hughes Medical Institute (HHMI). Wiedenheft is an HHMI fellow, and Lander a fellow with the Damon Runyon Cancer Research Foundation.
The microbial immune system can be likened to a vaccination program because of the adaptive-type nucleic acid-based line of defense deployed by a unit of DNA called CRISPR, which stands for Clustered Regularly Interspaced Short Palindromic Repeats. Although CRISPR defense systems are only found in microbes, they are analogous to the way in which the human immune system deploys short interfering RNAs (siRNAs) to fight off infections or correct genetic problems.
Usually located on a microbe’s chromosome, CRISPR units consist of “repeats,” base-pair sequences ranging from 30 to 60 nucleotides in length, that are separated by “spacers,” variable sequences, which are also 30 to 60 nucleotides in length. A microbe might harbor several CRISPR loci (sites) within its genome and each locus might contain between four and 100 CRISPR repeat-spacer units. When a microbe recognizes that it has been invaded, it incorporates a small piece of the invader’s DNA into one of its CRISPR units as a new spacer sequence.
“By integrating short fragments of foreign DNA into its CRISPR units, a microbe maintains a genetic record of all prior encounters with foreign transgressors,” says Doudna. “CRISPRs are transcribed and the long primary transcript is processed into a library of short CRISPR-derived RNAs (crRNAs), each of which contains a unique sequence that is complementary to a foreign nucleic acid challenger.”
In Escherichia coli, crRNAs are incorporated into the Cascade complex – Cascade stands for CRISPR-associated complex for antiviral defense. It is the mission of Cascade to detect and engage foreign DNA. Cascade will release crRNAs that will bind with foreign nucleic acid sequences – via base pair matching to a “seed” sequence of nucleotides – and silence or otherwise inactivate them. Cascade will also send out signals to recruit the enzyme Cas3 to join the battle. Cas3 is a single-stranded nuclease that can cleave foreign DNA into harmless pieces.
To learn how Cascade is able to carry out its mission, Doudna, Nogales, Wiedenheft, Lander and a team of colleagues determined the sub-nanometer structures of Cascade before and after binding to a target sequence of foreign DNA. They discovered Cascade’s seahorse-shaped architecture and found that crRNAs are displayed along the spine of the seahorse within a helical arrangement of protein subunits.
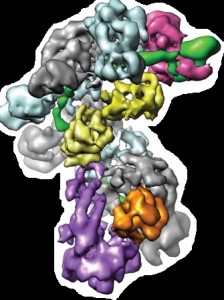
The architecture of the Cascade protein complex, a key player in the microbial immune system, resembles a seahorse, with crRNAs (green) displayed along the backbone within a helical arrangement of Cas protein subunits.
“The rigid backbone of this seahorse shaped architecture helps explain how the Cascade complex is able to accommodate crRNAs in a way that simultaneously protects them from degradation while maintaining their availability for base pairing to an invading nucleic acid target,” Wiedenheft says. “We further speculate that Cascade may pre-order a portion of the crRNA in a helical configuration and that this mechanism may be a structural solution for RNA-guided target binding that has been conserved through evolution.”
Although its seahorse shape is maintained throughout Cascade’s engagement with the enemy, the binding of the cRNAs to a foreign target does induce a “concerted conformational change” in the helical protein subunits running along Cascade’s backbone.
Says Nogales, “We speculate that this conformational change in the protein subunits generates a signal for recruiting Cas3 for further degradation of invading nucleic acid sequences.”
Cascade is a small complex by electron microscopy standards and its asymmetric conformation presented a major challenge that required the acquisition of a large amount of data during what Lander describes as “marathon” collection sessions.
“Three dimensional processing of electron microscopy data is generally a slow and iterative process, starting with very low resolution blobs that over time take shape and provide more intricate details,” says Lander. “Given Cascade’s seahorse shape, it was a bit like watching an embryo grow into a fully developed Cascade with a corkscrew spine. This is the very special kind of specimen that microscopists dream of.”
In addition to Doudna, Nogales, Wiedenheft and Lander, other co-authors of the Nature paper “Structures of the RNA-guided surveillance complex from a bacterial immune system” were Kaihong Zhou, Matthijs Jore, Stan Brouns and John van der Oost.
This research was supported by a grant from the National Science Foundation.
#
Lawrence Berkeley National Laboratory addresses the world’s most urgent scientific challenges by advancing sustainable energy, protecting human health, creating new materials, and revealing the origin and fate of the universe. Founded in 1931, Berkeley Lab’s scientific expertise has been recognized with 12 Nobel prizes. The University of California manages Berkeley Lab for the U.S. Department of Energy’s Office of Science. For more, visit www.lbl.gov.
Additional Information
For more information about Jennifer Doudna and her research group see http://rna.berkeley.edu/
For more information about Eva Nogales and her research group see http://cryoem.berkeley.edu/