Contact: Lynn Yarris (510) 486-5375, [email protected]
Scientists have identified a molecular cause behind the ravages of old age and in doing so have also shown how a natural process for fighting cancer in younger persons can actually promote cancer in older individuals. Cellular senescence, the process by which biological cells stop dividing in response to stress or damage to their DNA, was shown to trigger the secretion of proteins that cause inflammation in neighboring cells and tissue. Inflammation is linked to almost every major disease associated with aging, including many cancers.
Judith Campisi, a cell biologist who holds a joint appointment with the U.S. Department of Energy’s Lawrence Berkeley National Laboratory (Berkeley Lab) and the Buck Institute for Age Research, led this research.
“In this study, we provide for the first time a broad molecular description of how cellular senescence, which is well known as a mechanism for cancer prevention, also drives aging and age-related disease by changing the local tissue environment,” she said. “Our study also defines a new paradigm for how oncogenes promote and tumor suppressor genes slow down the development of cancer, and provides new evidence to support an evolutionary theory about aging.”
The results of this study were published on-line by the Public Library of Science (PLoS) in a paper entitled: “Senescence-Associated Secretory Phenotypes Reveal Cell-Nonautonomous Functions of Oncogenic RAS and the p53 Tumor Suppressor.”
Co-authoring the paper with Campisi were Jean-Philippe Coppé and Christopher Patil, members of Campisi’s research group in Berkeley Lab’s Life Sciences Division, Joshua Goldstein, now with the Novartis Research Foundation; Francis Rodier and Denise Muñoz of the Buck Institute; and Peter Nelson and Yu Sun from the Fred Hutchinson Cancer Research Center in Seattle.
The Good and the Bad of Cell Senescence
There are many different forms of cancer but all arise from cell division run amok. Nature has evolved two ways of shutting down cell-division and reining in uncontrolled growth: apoptosis, or cell suicide, and senescence, in which cells remain metabolically active but are no longer capable of dividing. Apoptosis as a tumor suppressor has been well characterized through in vivo studies, but the details of cellular senescence have not been so clearly defined. It was not until some 40 years after the process was first reported in cultures that an in vivo assay developed by Campisi and an earlier team of collaborators confirmed that it even occurs in living organisms.
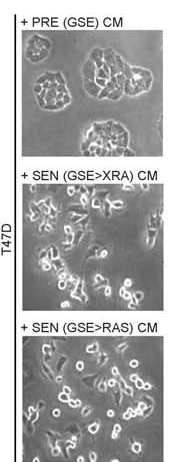
This trio of images show cancer cells that are normally non-aggressive and slow growing. The top image shows the cells in their typical state. In the middle image, the cells have lost the p53 tumor suppressor gene and are dividing much more aggressively. Cells in the final image have acquired the RAS oncogene and have started invading surrounding tissue.
Following her assay work, Campisi, a leading authority on cell senescence and the effects of aging, embarked upon studies in mice that led her to propose three years ago that with age an organism acquires enough senescent cells to create conditions in cells and tissue that are conducive to transforming benign lesions into malignant tumors. This hypothesis helped explain why tumor incidence increases exponentially with age.
“The evidence is increasing that genetic mutations alone are insufficient for cancer development,” Campisi said when she first announced this hypothesis. “Rather, malignant tumors also require a permissive tissue environment in which to develop and progress.”
In this latest research, she and her co-authors modified commercial antibody arrays to substantially improve their range and sensitivity, then measured the proteins secreted by senescent cells in culture, as well as from tissue samples that were collected from prostate cancer patients both before and after completing a DNA-damaging form of chemotherapy.
“We found that different types of cells from different tissue all secrete a remarkably similar set of proteins when they senesce in response to DNA-damaging radiation or chemotherapy,” Campisi said.
She and her co-authors branded this set of proteins as a senescence-associated secretory phenotype, or SASP, and determined that it occurs not only in cultured cells, but also in vivo in response to DNA damage.
“We showed that SASP develops slowly over several days and only after DNA damage of sufficient magnitude to induce senescence,” Campisi said. “All SASP features high levels of secreted inflammatory cytokines, immune modulators, and growth factors, which are associated with inflammation and malignancy.”
Oncogenes and Tumor Suppressors
While Campisi and her co-authors found that secretion of SASP was common to all senescent cells, there were distinct quantitative and qualitative differences among the different cell strains and lines. This paved the way for additional findings concerning genes that encode tumor promoting proteins (oncogenes) and those that encode tumor-suppressing proteins. For example, normal cells that acquired a highly active version of the RAS oncogene became senescent and secreted higher levels of SASP components. This very intense SASP also happened when cells lost the functions of the p53 tumor suppressor gene.
“Our findings help explain why senescent cells promote the growth and aggressiveness of nearby pre-cancerous or cancer cells, and define a new mechanism by which pre-cancerous or cancer cells that have lost the p53 tumor suppressor gene, or gained an oncogene such as RAS, promote cancer so efficiently,” Campisi said.

Judith Campisi, a cell biologist who holds a joint appointment with Berkeley Lab and the Buck Institute, is a leading authority on cell senescence and the effects of aging. (Photo by Roy Kaltschmidt, Berkeley Lab Public Affairs)
These findings also raise a cautionary note with regards to the use of DNA-damaging chemotherapy on patients under the age of 50. Such treatments are aimed at cells that are undergoing rapid division but impact neighboring normal cells as well. As the findings of Campisi and her co-authors show, sufficient DNA damage forces both cancerous and normal cells into senescence, releasing SASP components. The resulting inflammation of neighboring cells and tissue would explain why so many patients receiving chemotherapy experience flu-like symptoms during treatments. Such inflammation can also promote the development of further cancers later in life.
“Chemotherapy can cure cancer but our results suggest that this cure comes with a high cost and poses a risk that could come back to haunt the patient,” Campisi said. “There is a great need for new biological therapies that more effectively exploit the specific differences between normal and cancer cells. Meanwhile, prevention remains the best treatment for cancer.”
Evolutionary Implications
The findings by Campisi and her co-authors that cellular senescence can be beneficial to the young and deleterious to the old provide what may be the first molecular evidence in support of a major evolutionary theory on aging called “antagonistic pleiotropy.” This theory holds that natural selection can favor a trait that contributes to early life fitness even if that trait is harmful in older individuals.
“The dynamic we have shown between the cancer prevention and aging effects of cellular senescence is an example of the trade-offs between early fitness and late life survival, exactly what is predicted by antagonistic pleiotropy,” said Campisi. “The challenge now is to preserve the anti-cancer activity of cellular senescence while dampening its pro-aging effects. Although senescent cells exist for the good purpose of preventing cancer, we don’t want them to hang around – we want the body to be able to get rid of them.”
To this end, Campisi and her colleagues are now developing mouse models through which they can determine whether there are molecular pathways by which senescent cells can either be safely expelled, or selectively destroyed.
“Senescent cells are extremely stable and do not die, therefore they accumulate in the body over time in multiple tissues,” she said. “The true test as to whether cellular senescence is driving the pathologies associated with aging will come if we are able to get rid of senescent cells in our mouse model.”
This work was supported by grants from the National Institutes of Health.
Berkeley Lab is a U.S. Department of Energy national laboratory located in Berkeley, California. It conducts unclassified scientific research and is managed by the University of California. Visit our Website at www.lbl.gov
Additional Information
- For more information on the research of Judy Campisi visit her Website at http://www.lbl.gov/lifesciences/labs/campisi_lab.html
- For more information on the Buck Institute, the only freestanding institute in the United States that is dedicated to basic research on aging and age-associated disease, visit the Website at www.buckinstitute.org