Scientists from the U.S. Department of Energy’s Lawrence Berkeley National Laboratory have learned how an interval of DNA in an unexplored region of the human genome increases the risk for coronary artery disease, the leading cause of death worldwide.
Their research paints a fuller picture of a genetic risk for the disease that was discovered only three years ago and which lurks in one out of two people.
It also reinforces the tantalizing possibility that many more disease risks — and potential disease-fighting therapies — are hidden in the vast and uncharted part of the genome that doesn’t contain instructions for making proteins.

A genetic risk for heart disease comes into focus. Berkeley Lab's Len Pennacchio (left) and Axel Visel are among a team of scientists who have learned how an interval of DNA in a region of the human genome that doesn't code for proteins increases the risk for coronary artery disease, the leading cause of death worldwide.
The research is reported in the February 21 advance online publication of the journal Nature.
The team focused on an interval of DNA in chromosome 9p21. People who carry variations of this interval have an increased chance of developing coronary artery disease, which is an accumulation of plaque in coronary arteries that restricts blood flow to the heart and causes heart attacks.
Determining how this DNA contributes to the disease is difficult because it’s in the poorly understood part of the genome that doesn’t code for proteins, the workhorses of cellular function.
In groundbreaking research, the Berkeley Lab scientists found that the DNA interval regulates a pair of genes that inhibit cell division, and that bad copies of the interval reduce the genes’ expression. Although more work is needed to understand how this mechanism contributes to coronary artery disease, the researchers speculate that the hobbled genes allow vascular cells to proliferate unchecked and narrow coronary arteries.
“We show that this non-coding interval affects the expression of two cell cycle inhibitor genes located almost 100,000 base pairs away. We believe that something goes awry in variants of this interval, causing vascular cells to divide and multiply more quickly than usual,” says Len Pennacchio, a geneticist with Berkeley Lab’s Genomics Division who conducted the research with Axel Visel and several other scientists from Berkeley Lab, as well as Jonathan Cohen of the University of Texas Southwestern Medical Center.
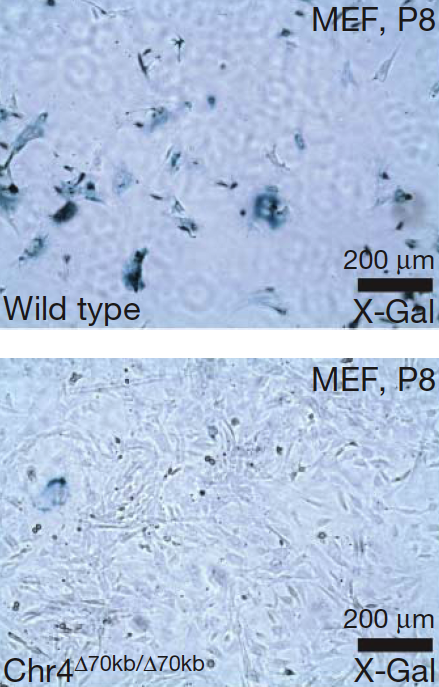
The way in which cells proliferate is disrupted when the interval of DNA that increases the risk for coronary artery disease is removed, as seen in cultures of these mouse embryonic fibroblasts. In the bottom image, cells from "knock-out" mice fail to become senescent -- or lose the ability to divide -- as evident from the absence of senescence staining. In the top image, cells from normal mice become senescent. The scientists speculate that the risk interval may cause vascular cells to proliferate unchecked and narrow coronary arteries.
The link between an interval of DNA in chromosome 9p21 and a risk for coronary artery disease was established in several recent studies, one of which was published in the journal Science in 2007. In that study, led by Cohen and co-authored by several scientists including Pennacchio, the researchers scoured the human genome for differences in people who have coronary artery disease versus people who don’t.
This genome-wide association analysis alighted on a stretch of DNA in chromosome 9p21 that spans 58,000 base pairs of DNA. The study found that people with bad copies of this interval have a moderately higher risk of developing coronary artery disease. In addition, 50 percent of people have one bad copy and 25 percent have two bad copies.
“The risk of coronary artery disease isn’t very high in any give person with bad copies. But they are so common that population-wide the effect is significant,” says Pennacchio.
Remarkably, the study also found that the DNA interval isn’t associated with known risks for coronary artery disease such as diabetes, high blood pressure, and high cholesterol level. An unknown mechanism was at work.
“We landed on this risk interval and immediately said ‘wow!’ why doesn’t it link to problems that we know cause coronary artery disease?” says Pennacchio. “So the big question became: what is this DNA doing?”
Adding to the mystery, the DNA interval is among the 98 percent of our genome that doesn’t code for proteins. Most efforts to determine the function of the genome have focused on the two percent of our DNA that overlaps protein-coding genes. Scientists are just now beginning to explore the non-coding region, once referred to as “junk DNA.”
As part of this effort, the Berkeley Lab scientists set out to determine the function of the DNA interval in chromosome 9p21 that’s linked to coronary artery disease. They removed an analogous section of DNA from the genomes of mice, then tracked what happened.
The expression level of two genes located far away, Cdkn2a and Cdkn2b, plummeted by about 90 percent in the “knock-out” mice compared to normal mice. These genes are important in controlling cell cycles and have been linked to cancer when mutated, but they had never been linked to coronary artery disease.
The scientists also studied heart tissue of the “knock-out” mice and found that the smooth muscle cells from their aortas had increased proliferation, a hallmark of coronary artery disease.
“Our research shows that the DNA interval plays a pivotal role in regulating the expression of two genes that control cell cycles. It also suggests that variants of the interval spur the progression of coronary artery disease by altering the dynamics of vascular cells,” says Pennacchio.
With this mechanism identified, scientists can develop therapies that fight coronary artery disease by targeting the two genes and jumpstarting them into action, says Pennacchio. He also believes that the genetic roots of many other diseases will be unearthed as scientists learn how to decipher the function of non-coding DNA.
“Non-coding DNA is a huge area of the genome, waiting to be explored, which could have huge dividends for understanding and treating disease,” says Pennacchio.
The research was funded by the National Institutes of Health.
Other Berkeley Lab scientists involved in the research include Yiwen Zhu, Dalit May, Veena Afzal, Elaine Gong, Cattia Attanasio, Matthew Blow, and Eddy Rubin.
Berkeley Lab is a U.S. Department of Energy national laboratory located in Berkeley, California. It conducts unclassified scientific research for DOE’s Office of Science and is managed by the University of California. Visit our website at http://www.lbl.gov.