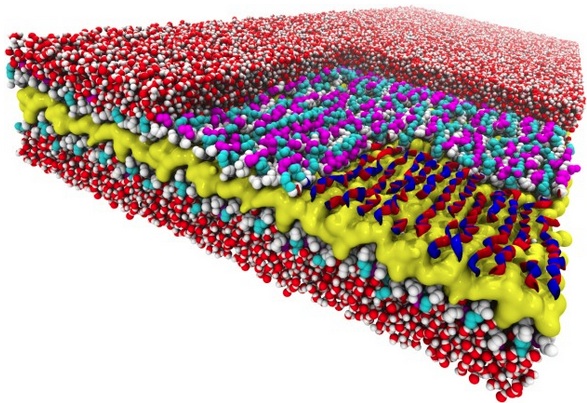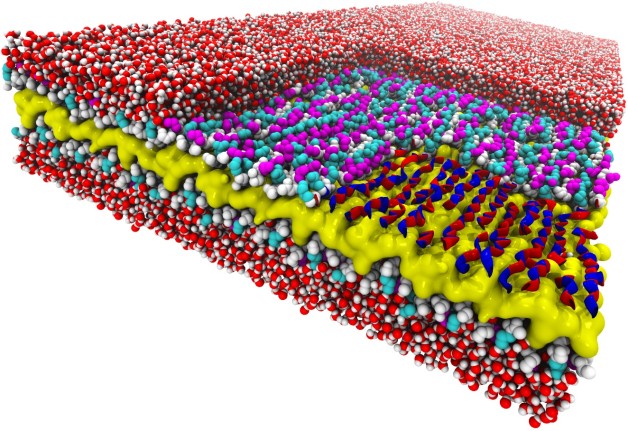
Snakes on a plane: This atomic-resolution simulation of a two-dimensional peptoid nanosheet reveals a snake-like structure never seen before. The nanosheet’s layers include a water-repelling core (yellow), peptoid backbones (white), and charged sidechains (magenta and cyan). The right corner of the top layer of the nanosheet has been “removed” to show how the backbone’s alternating rotational states give the backbones a snake-like appearance (red and blue ribbons). Surrounding water molecules are red and white. (Credit: Ranjan Mannige, Berkeley Lab)
Scientists aspire to build nanostructures that mimic the complexity and function of nature’s proteins, but are made of durable and synthetic materials. These microscopic widgets could be customized into incredibly sensitive chemical detectors or long-lasting catalysts, to name a few possible applications.
But as with any craft that requires extreme precision, researchers must first learn how to finesse the materials they’ll use to build these structures. A discovery by scientists from the Department of Energy’s Lawrence Berkeley National Laboratory (Berkeley Lab), and reported Oct. 7 in the advance online publication of the journal Nature, is a big step in this direction.
The scientists discovered a design rule that enables a recently created material to exist. The material is a peptoid nanosheet. It’s a flat structure only two molecules thick, and it’s composed of peptoids, which are synthetic polymers closely related to protein-forming peptides.
The design rule controls the way in which polymers adjoin to form the backbones that run the length of nanosheets. Surprisingly, these molecules link together in a counter-rotating pattern not seen in nature. This pattern allows the backbones to remain linear and untwisted, a trait that makes peptoid nanosheets larger and flatter than any biological structure.
The Berkeley Lab scientists say this never-before-seen design rule could be used to piece together complex nanosheet structures and other peptoid assemblies such as nanotubes and crystalline solids.
What’s more, they discovered it by combining computer simulations with x-ray scattering and imaging methods to determine, for the first time, the atomic-resolution structure of peptoid nanosheets.
“This research suggests new ways to design biomimetic structures,” says Steve Whitelam, a co-corresponding author of the Nature paper. “We can begin thinking about using design principles other than those nature offers.”
Whitelam is a staff scientist in the Theory Facility at the Molecular Foundry, a DOE Office of Science user facility located at Berkeley Lab. He led the research with co-corresponding author Ranjan Mannige, a postdoctoral researcher at the Molecular Foundry; and Ron Zuckermann, who directs the Molecular Foundry’s Biological Nanostructures Facility. They used the high-performance computing resources of the National Energy Research Scientific Computing Center (NERSC), another DOE Office of Science user facility located at Berkeley Lab.

The Molecular Foundry scientists who helped discover a new nano design rule. From left, Ellen Robertson, Alessia Battigelli, Ron Zuckermann, Caroline Proulx, Stephen Whitelam, and Ranjan Mannige. (Credit: Roy Kaltschmidt, Berkeley Lab)
Peptoid nanosheets were discovered by Zuckermann’s group five years ago. They found that under the right conditions, peptoids self assemble into two-dimensional assemblies that can grow hundreds of microns across. This “molecular paper” has become a hot prospect as a protein-mimicking platform for molecular design.
To learn more about this potential building material, the scientists set out to learn its atom-resolution structure. This involved feedback between experiment and theory. Microscopy and scattering data gathered at the Molecular Foundry and the Advanced Light Source, also a DOE Office of Science user facility located at Berkeley Lab, were compared with molecular dynamics simulations conducted at NERSC.
The research revealed several new things about peptoid nanosheets. Their molecular makeup varies throughout their structure, they can be formed only from peptoids of a certain minimum length, they contain water pockets, and they are potentially porous when it comes to water and ions.
These insights are intriguing on their own, but when the scientists examined the structure of the nanosheets’ backbone, they were surprised to see a design rule not found in the field of protein structural biology.
Here’s the difference: In nature, proteins are composed of beta sheets and alpha helices. These fundamental building blocks are themselves composed of backbones, and the polymers that make up these backbones are all joined together using the same rule. Each adjacent polymer rotates incrementally in the same direction, so that a twist runs along the backbone.
This rule doesn’t apply to peptoid nanosheets. Along their backbones, adjacent monomer units rotate in opposite directions. These counter-rotations cancel each other out, resulting in a linear and untwisted backbone. This enables backbones to be tiled in two dimensions and extended into large sheets that are flatter than anything nature can produce.
“It was a big surprise to find the design rule that makes peptoid nanosheets possible has eluded the field of biology until now,” says Mannige. “This rule could perhaps be used to build many more unrealized structures.”
Adds Zuckermann, “We also expect there are other design principles waiting to be discovered, which could lead to even more biomimetic nanostructures.”
A simulation of a peptoid nanosheet, shown first from a top-down view with the peptoid backbones colored to highlight their snake-like structure. The view then rotates to the side, and finally transitions to an all-atom representation. (Credit: Ron Zuckermann and Ranjan Mannige, Berkeley Lab)
Other Molecular Foundry scientists who contributed to this research are Thomas Haxton, Caroline Proulx, Ellen Robertson, and Alessia Battigelli.
This research was conducted at the Molecular Foundry, a DOE Office of Science user facility located at Berkeley Lab. The work was supported by the Defense Threat Reduction Agency, with additional funding provided by the Natural Sciences and Engineering Research Council of Canada. Part of this research was carried out through a User Project at the Molecular Foundry led by New York University’s Glenn Butterfoss.
###
Lawrence Berkeley National Laboratory addresses the world’s most urgent scientific challenges by advancing sustainable energy, protecting human health, creating new materials, and revealing the origin and fate of the universe. Founded in 1931, Berkeley Lab’s scientific expertise has been recognized with 13 Nobel prizes. The University of California manages Berkeley Lab for the U.S. Department of Energy’s Office of Science. For more, visit www.lbl.gov.
DOE’s Office of Science is the single largest supporter of basic research in the physical sciences in the United States, and is working to address some of the most pressing challenges of our time. For more information, please visit science.energy.gov.
Additional information:
- A paper describing this research, entitled “Peptoid nanosheets exhibit a new secondary-structure motif,” was published Oct. 7, 2015, in the advance online publication of the journal Nature.