The same electrostatic charge that can make hair stand on end and attach balloons to clothing could be an efficient way to drive atomically thin electronic memory devices of the future, according to a new study led by researchers at the Department of Energy’s Lawrence Berkeley National Laboratory (Berkeley Lab).
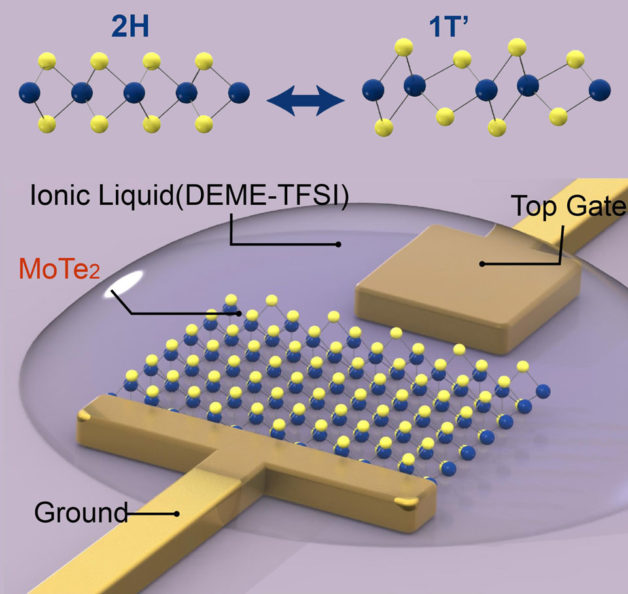
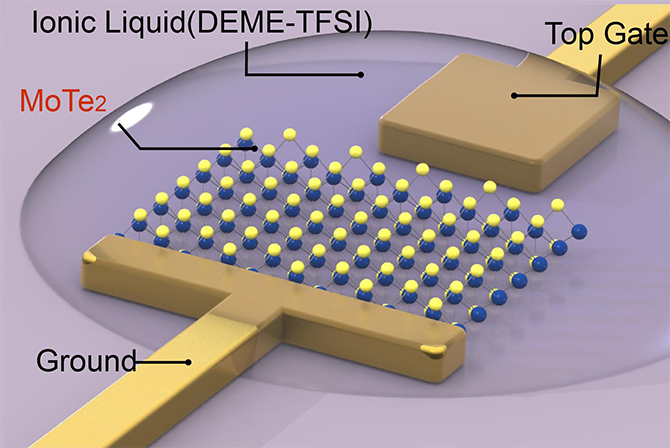
Schematic shows the configuration for structural phase transition on a molybdenum ditelluride monolayer (MoTe2, shown as yellow and blue spheres), which is anchored by a metal electrodes (top gate and ground). The ionic liquid covering the monolayer and electrodes enables a high density of electrons to populate the monolayer, leading to changes in the structural lattice from a hexagonal (2H) to monoclinic (1T’) pattern. (Credit: Ying Wang/Berkeley Lab)
In a study published today in the journal Nature, scientists have found a way to reversibly change the atomic structure of a 2-D material by injecting, or “doping,” it with electrons. The process uses far less energy than current methods for changing the configuration of a material’s structure.
“We show, for the first time, that it is possible to inject electrons to drive structural phase changes in materials,” said study principal investigator Xiang Zhang, senior faculty scientist at Berkeley Lab’s Materials Sciences Division and a professor at UC Berkeley. “By adding electrons into a material, the overall energy goes up and will tip off the balance, resulting in the atomic structure rearranging to a new pattern that is more stable. Such electron doping-driven structural phase transitions at the 2-D limit is not only important in fundamental physics; it also opens the door for new electronic memory and low-power switching in the next generation of ultra-thin devices.”
Switching a material’s structural configuration from one phase to another is the fundamental, binary characteristic that underlies today’s digital circuitry. Electronic components capable of this phase transition have shrunk down to paper-thin sizes, but they are still considered to be bulk, 3-D layers by scientists. By comparison, 2-D monolayer materials are composed of a single layer of atoms or molecules whose thickness is 100,000 times as small as a human hair.
“The idea of electron doping to alter a material’s atomic structure is unique to 2-D materials, which are much more electrically tunable compared with 3-D bulk materials,” said study co-lead author Jun Xiao, a graduate student in Zhang’s lab.
The classic approach to driving the structural transition of materials involves heating to above 500 degrees Celsius. Such methods are energy-intensive and not feasible for practical applications. In addition, the excess heat can significantly reduce the life span of components in integrated circuits.
A number of research groups have also investigated the use of chemicals to alter the configuration of atoms in semiconductor materials, but that process is still difficult to control and has not been widely adopted by industry.
“Here we use electrostatic doping to control the atomic configuration of a two-dimensional material,” said study co-lead author Ying Wang, another graduate student in Zhang’s lab. “Compared to the use of chemicals, our method is reversible and free of impurities. It has greater potential for integration into the manufacturing of cell phones, computers, and other electronic devices.”
The researchers used molybdenum ditelluride (MoTe2), a typical 2-D semiconductor, and coated it with an ionic liquid (DEME-TFSI), which has an ultra-high capacitance, or ability to store electric charges. The layer of ionic liquid allowed the researchers to inject the semiconductor with electrons at a density of a hundred trillion to a quadrillion per square centimeter. It is an electron density that is one to two orders higher in magnitude than what could be achieved in 3-D bulk materials, the researchers said.
Through spectroscopic analysis, the researchers determined that the injection of electrons changed the atoms’ arrangement of the molybdenum ditelluride from a hexagonal shape to one that is monoclinic, which has more of a slanted cuboid shape. Once the electrons were retracted, the crystal structure returned to its original hexagonal pattern, showing that the phase transition is reversible. Moreover, these two types of atom arrangements have very different symmetries, providing a large contrast for applications in optical components.
“Such an atomically thin device could have dual functions, serving simultaneously as optical or electrical transistors, and hence broaden the functionalities of the electronics used in our daily lives,” said Wang.
This work was supported by DOE’s Office of Science and by the National Science Foundation.
###
Lawrence Berkeley National Laboratory addresses the world’s most urgent scientific challenges by advancing sustainable energy, protecting human health, creating new materials, and revealing the origin and fate of the universe. Founded in 1931, Berkeley Lab’s scientific expertise has been recognized with 13 Nobel Prizes. The University of California manages Berkeley Lab for the U.S. Department of Energy’s Office of Science. For more, visit www.lbl.gov.
DOE’s Office of Science is the single largest supporter of basic research in the physical sciences in the United States, and is working to address some of the most pressing challenges of our time. For more information, please visit science.energy.gov.