Researchers at DOE’s Lawrence Berkeley National Laboratory (Berkeley Lab) have 3D-printed an all-liquid device that, with the click of a button, can be repeatedly reconfigured on demand to serve a wide range of applications – from making battery materials to screening drug candidates.

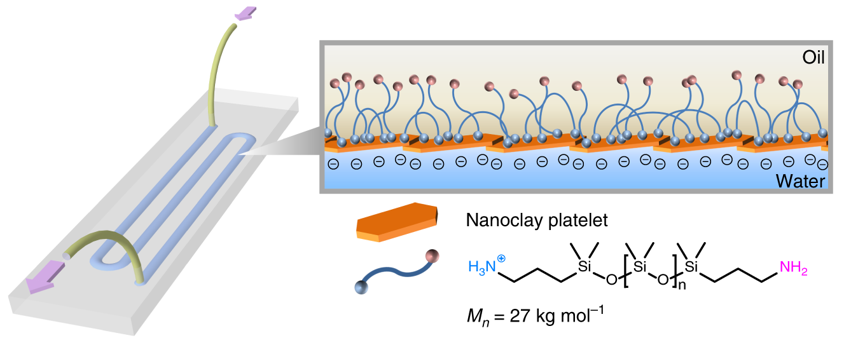
The 3D-printed all-liquid device can be programmed to function like an artificial circulatory system that separates molecules flowing through the channel and automatically removes unwanted byproducts. (Credit: Berkeley Lab)
“What we demonstrated is remarkable. Our 3D-printed device can be programmed to carry out multistep, complex chemical reactions on demand,” said Brett Helms, a staff scientist in Berkeley Lab’s Materials Sciences Division and Molecular Foundry, who led the study. “What’s even more amazing is that this versatile platform can be reconfigured to efficiently and precisely combine molecules to form very specific products, such as organic battery materials.”
The study’s findings, which were reported in the journal Nature Communications, is the latest in a series of experiments at Berkeley Lab that fabricate all-liquid materials with a 3D printer.
Last year, a study co-authored by Helms and Thomas Russell, a visiting researcher from the University of Massachusetts at Amherst who leads the Adaptive Interfacial Assemblies Toward Structured Liquids Program in Berkeley Lab’s Materials Sciences Division, pioneered a new technique for printing various liquid structures – from droplets to swirling threads of liquid – within another liquid.
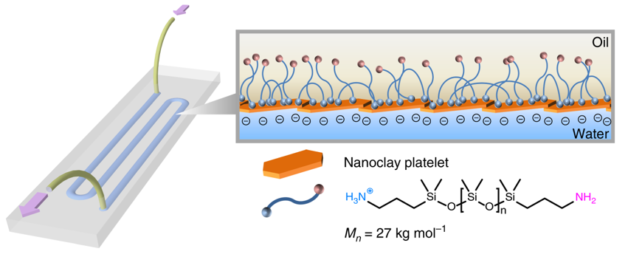
When two liquids – one containing nanoscale clay particles, another containing polymer particles – are printed onto a glass substrate, they come together at the interface of the two liquids and within milliseconds form a very thin channel or tube about 1 millimeter in diameter. (Credit: Berkeley Lab)
“After that successful demonstration, a bunch of us got together to brainstorm on how we could use liquid printing to fabricate a functioning device,” said Helms. “Then it occurred to us: If we can print liquids in defined channels and flow contents through them without destroying them, then we could make useful fluidic devices for a wide range of applications, from new types of miniaturized chemical laboratories to even batteries and electronic devices.”
To make the 3D-printable fluidic device, lead author Wenqian Feng, a postdoctoral researcher in Berkeley Lab’s Materials Sciences Division, designed a specially patterned glass substrate. When two liquids – one containing nanoscale clay particles, another containing polymer particles – are printed onto the substrate, they come together at the interface of the two liquids and within milliseconds form a very thin channel or tube about 1 millimeter in diameter.
VIDEO: Researchers from Berkeley Lab’s Materials Sciences Division and Molecular Foundry have 3D-printed an all-liquid “lab on a chip” that can serve a wide range of applications, from making battery materials to screening drug candidates. (Credit: Helms Research Group/Berkeley Lab)
Once the channels are formed, catalysts can be placed in different channels of the device. The user can then 3D-print bridges between channels, connecting them so that a chemical flowing through them encounters catalysts in a specific order, setting off a cascade of chemical reactions to make specific chemical compounds. And when controlled by a computer, this complex process can be automated “to execute tasks associated with catalyst placement, build liquid bridges within the device, and run reaction sequences needed to make molecules,” said Russell.
The multitasking device can also be programmed to function like an artificial circulatory system that separates molecules flowing through the channel and automatically removes unwanted byproducts while it continues to print a sequence of bridges to specific catalysts, and carry out the steps of chemical synthesis.
“The form and functions of these devices are only limited by the imagination of the researcher,” explained Helms. “Autonomous synthesis is an emerging area of interest in the chemistry and materials communities, and our technique for 3D-printing devices for all-liquid flow chemistry could help to play an important role in establishing the field.”
Added Russell: “The combination of materials science and chemistry expertise at Berkeley Lab, along with world-class user facilities available to researchers from all over the world, and the young talent that is drawn to the Lab is unique. We couldn’t have developed this program anywhere else.”
The researchers next plan to electrify the walls of the device using conductive nanoparticles to expand the types of reactions that can be explored. “With our technique, we think it should also be possible to create all-liquid circuitry, fuel cells, and even batteries,” said Helms. “It’s been really exciting for our team to combine fluidics and flow chemistry in a way that is both user-friendly and user-programmable.”
The Molecular Foundry is a DOE Office of Science User Facility that specializes in nanoscale science.
Researchers from UC Berkeley also contributed to the study.
The technology is available for licensing and collaboration. If interested, please contact Berkeley Lab’s Intellectual Property Office, [email protected].
This work was supported by the DOE Office of Science.
###
Founded in 1931 on the belief that the biggest scientific challenges are best addressed by teams, Lawrence Berkeley National Laboratory and its scientists have been recognized with 13 Nobel Prizes. Today, Berkeley Lab researchers develop sustainable energy and environmental solutions, create useful new materials, advance the frontiers of computing, and probe the mysteries of life, matter, and the universe. Scientists from around the world rely on the Lab’s facilities for their own discovery science. Berkeley Lab is a multiprogram national laboratory, managed by the University of California for the U.S. Department of Energy’s Office of Science.
DOE’s Office of Science is the single largest supporter of basic research in the physical sciences in the United States, and is working to address some of the most pressing challenges of our time. For more information, please visit science.energy.gov.