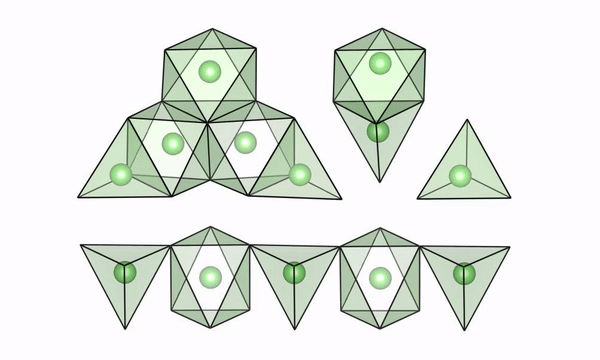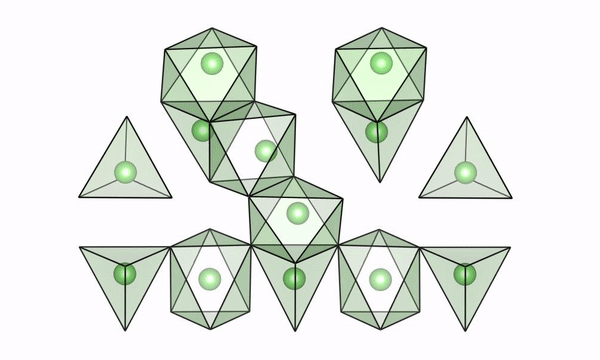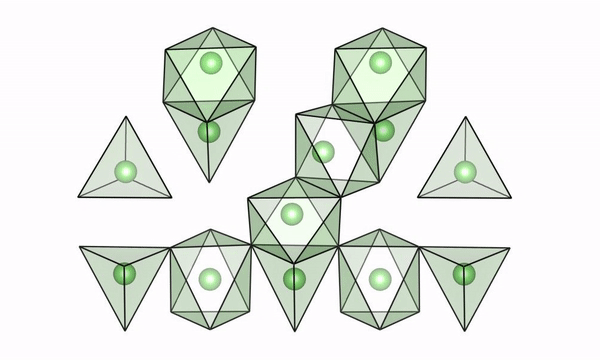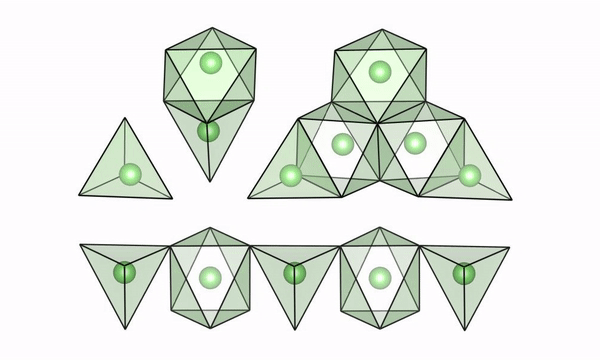
A movie of lithium ions quickly moving along “easy pathways” in intermediate configurations of LTO (lithium titanate). (Credit: Brookhaven National Laboratory)
Berkeley Lab researchers, working with a team at Brookhaven National Laboratory, have made a key discovery about the dynamic structural changes in a material called lithium titanate, putting scientists one step closer to achieving a fast-charging lithium battery.
The scientists used both experimental and computational techniques to examine lithium titanate, or LTO, while it was undergoing fast charge and were able to see why lithium ions are able to move so fast. Their findings were published recently in the journal Science.
“We were able to visualize a key feature of the structure of LTO,” said Tina Chen, a UC Berkeley graduate student and Berkeley Lab researcher who worked with Berkeley Lab scientist Gerbrand Ceder on the study. “We believe that if we can find materials that can accommodate this feature when they’re cycling, then they may also be good candidates to have high rate capability. LTO would serve as the anode in a battery, but we also want to find better materials for the cathode, and potentially we could apply this to solid electrolytes with higher lithium mobility.”
Lithium-ion batteries, such as those used in cell phones and electric vehicles, can take hours to charge. “There are a lot of materials that if you try to charge your battery too quickly then the capacity will drastically decrease,” Chen said. “The goal is to be able to charge your battery very quickly and still maintain a high capacity.”
Click here for a news release from Brookhaven National Laboratory on this study.