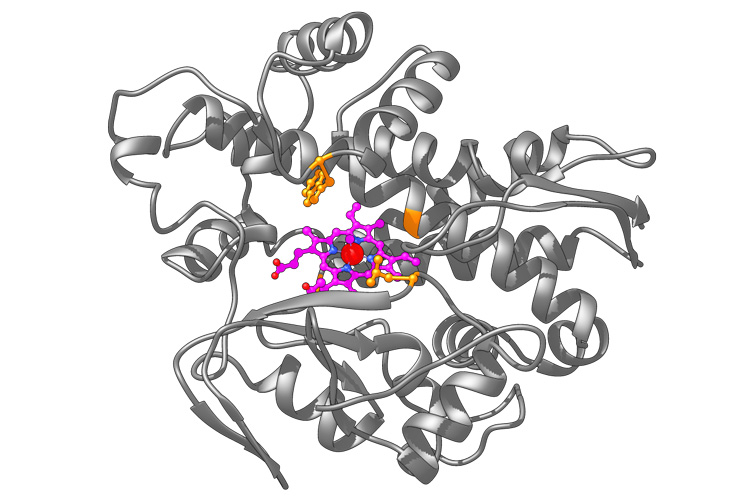
An artificial metalloenzyme based on the natural enzyme called P450 (gray structure). UC Berkeley chemists created a heme molecule (magenta) with an embedded iridium atom (red) that, in E. coli, was incorporated into P450 to execute a reaction unknown in the natural world. (UC Berkeley image by Brandon Bloomer)
Adapted from a UC Berkeley news release.
Synthetic biologists have successfully engineered microbes to make chemicals cheaply and more sustainably. However, researchers have been limited by the fact that microbes can only make molecules using chemical reactions seen in nature.
A collaboration between scientists at Berkeley Lab and UC Berkeley has engineered the microbe E. coli to produce a molecule that, until now, could only be synthesized in a laboratory.
To achieve this outcome, the researchers integrated a specific type of modified enzyme into E. coli, along with a pathway to produce a precursor molecule. This was then converted into a novel product from a reaction not previously seen in nature.
This research opens the door to the production of a wide range of chemicals from microbes, the researchers said. Co-author Aindrila Mukhopadhyay, Berkeley Lab senior scientist in the Biosciences Area and vice president of the Biofuels and Bioproducts Division at the Joint BioEnergy Institute (JBEI) said this method could be a game changer in terms of microbial production to make pharmaceuticals, as well as sustainable fuels.
“Today, many drugs are laboriously extracted from biological systems that are challenging to cultivate or processes that negatively impact the environment. To be able to reliably make these compounds in a lab using biotechnology would really address a lot of these problems,” she said.
This applies to making “not just medicines, but precursors to polymers, renewable plastics, biofuels, building materials, the whole gamut of things that we use today, from detergents to lubricants to paints to pigments to fabric,” Mukhopadhyay added. “We really need disruptive new technologies, and this most definitely is one of them.”
In addition to Mukhopadhyay, Berkeley Lab co-authors of the study included the Biosciences Area’s Jing Huang, Douglas S. Clark, and Jay D. Keasling and the Chemical Sciences Division’s Zhennan Liu, Brandon J. Bloomer, and John F. Hartwig.
JBEI is a U.S. Department of Energy (DOE) Bioenergy Research Center led by Berkeley Lab.