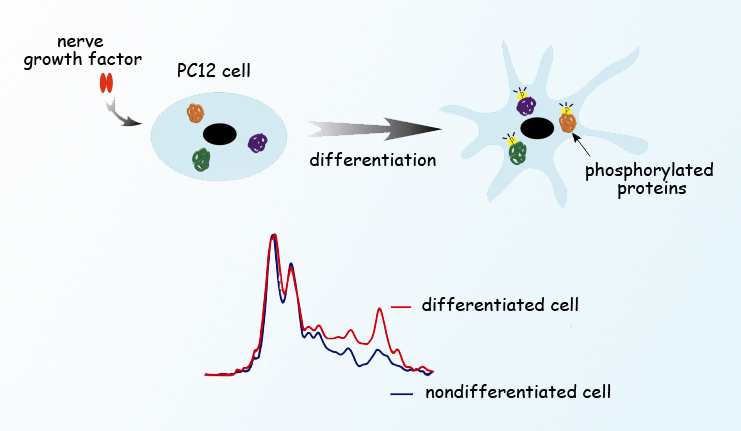
Berkeley Lab scientists observed phosphorylation in living PC12 cells stimulated by nerve growth factor as they differentiated and sent out neuron-like neurites. The researchers imaged individual cells and simultaneously obtained absorption spectra using synchrotron radiation from the Advanced Light Source. Cells not stimulated with nerve growth factor did not differentiate and showed different infrared absorption spectra.
Knowing how a living cell works means knowing how the chemistry inside the cell changes as the functions of the cell change. Protein phosphorylation, for example, controls everything from cell proliferation to differentiation to metabolism to signaling, and even programmed cell death (apoptosis), in cells from bacteria to humans. It’s a chemical process that has long been intensively studied, not least in hopes of treating or eliminating a wide range of diseases. But until now the close-up view – watching phosphorylation work at the molecular level as individual cells change over time – has been impossible without damaging the cells or interfering with the very processes that are being examined.
“To look into phosphorylation, researchers have labeled specific phosphorylated proteins with antibodies that carry fluorescent dyes,” says Hoi-Ying Holman of the U.S. Department of Energy’s Lawrence Berkeley National Laboratory (Berkeley Lab). “That gives you a great image, but you have to know exactly what to label before you can even begin.”
Holman and her coworkers worked with colleagues from the San Diego and Berkeley campuses of the University of California to develop a new technique for monitoring protein phosphorylation inside single living cells, tracking them over a week’s time as they underwent a series of major changes.
“Now we can follow cellular chemical changes without preconceived notions of what they might be,” says Holman, a pioneer in infrared (IR) studies of living cells who is director of the Berkeley Synchrotron Infrared Structural Biology program at Berkeley Lab’s Advanced Light Source (ALS) and head of the Chemical Ecology Research group in the Earth Sciences Division . “We’ve monitored unlabeled living cells by studying the nonperturbing absorption of a wide spectrum of bright synchrotron infrared radiation from the ALS.”
The researchers report their results in the American Chemical Society journal Analytical Chemistry.
Phosphorylation fundamentals
Phosphorylating enzymes add one or more phosphate groups to three amino-acid residues common in proteins – serine, threonine, or tyrosine – which activates the proteins; removing the phosphate reverses the process. The research goal is to learn exactly when proteins such as enzymes and receptors are switched on and off by phosphorylation, and which cells within a population are responding to cause specific changes – for example, during differentiation of a progenitor cell into its functional form.
To avoid killing cells or introducing modified proteins or foreign bodies that may alter their behavior, scientists can use a method called Fourier-transform infrared (FTIR) spectromicroscopy; because infrared light has lower photon energy than x-rays, it can peer inside living cells without damaging them. Different components and different states of the cell absorb different wavelengths of the broad infrared spectrum; applying the Fourier-transform algorithm allows signals of all frequencies to be recorded simultaneously, pinpointing when, where, and what chemical changes are occurring.
Most infrared sources are dim, however, so the information from typical IR set-ups is limited in resolution and has a low signal-to-noise ratio. Infrared from the ALS’s synchrotron light source is a hundred to a thousand times brighter.
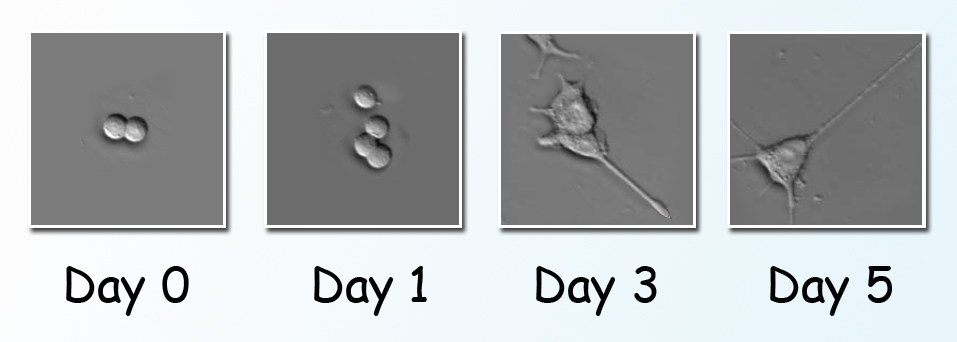
PC12 cells treated with nerve growth factor underwent a series of changes due to phosphorylation. Beginning at Day 3 they sent out neurites, resembling the growth of nerve cells. Spectromicroscopy at beamline 1.4.3 of the Advanced Light Source tracked specific local chemical changes in the living cells.
Previously Holman and her colleagues have used IR beamline 1.4.3, managed by Berkeley Lab’s Michael Martin and Hans Bechtel, to obtain spectra from living organisms in rock, soil, and water. They have monitored ongoing biochemistry within living bacteria adapting to stress, and more recently within individual skin connective tissue cells (fibroblasts) from patients with mitochondrial disorders. (Mitochondria are the cellular organelles commonly known as the “power-plants” of the cell.)
The present study was done with a line of cultured cells called PC12. When nerve growth factor, a small protein, is introduced into a PC12 cell, the cell begins to send out neurites resembling the projections from nerve cell bodies. Although originally derived from a tumor of the rat’s adrenal gland, PC12 has become, rather counterintuitively, a valuable model of how nerve cells differentiate from their unspecialized progenitors.
Berkeley Lab postdoctoral fellow Liang Chen began the current experiments by introducing nerve growth factor to groups of PC12 cells to induce them to differentiate; one group of cells was left untreated as a control. The cells were cultured on gold-coated slides in chambers maintained at body temperature in a humidified environment and supplied with nutrients. Individual cells of a group were positioned under the infrared beam at the beamline 1.4.3 endstation.
FTIR spectra were collected before and after the nerve growth factor was introduced. After stimulation, the spectra were taken first at short intervals, from two to sixty minutes apart. Additional spectra were collected of cells in other groups on the third, fifth, and seventh day of continued stimulation.
The first day’s spectra revealed spikes in phosphorylation activity within minutes after the addition of the nerve growth factor, in concert with changes in the ratios of such important chemical contents of the cell as proteins, carbohydrates, and lipids. Phosphorylation subsequently waned, then picked up again in another burst of activity on Day 3, just as the cells began to extend neurites.
By comparing results with quantum chemistry simulations by Berkeley Lab’s Zhao Hao — predicting what should be observed from first principles — as well as with results from partial studies using other methods, the researchers confirmed the monitoring of phosphorylation phases, their timing, and their target proteins, along with associated changes in other substances in the cell.
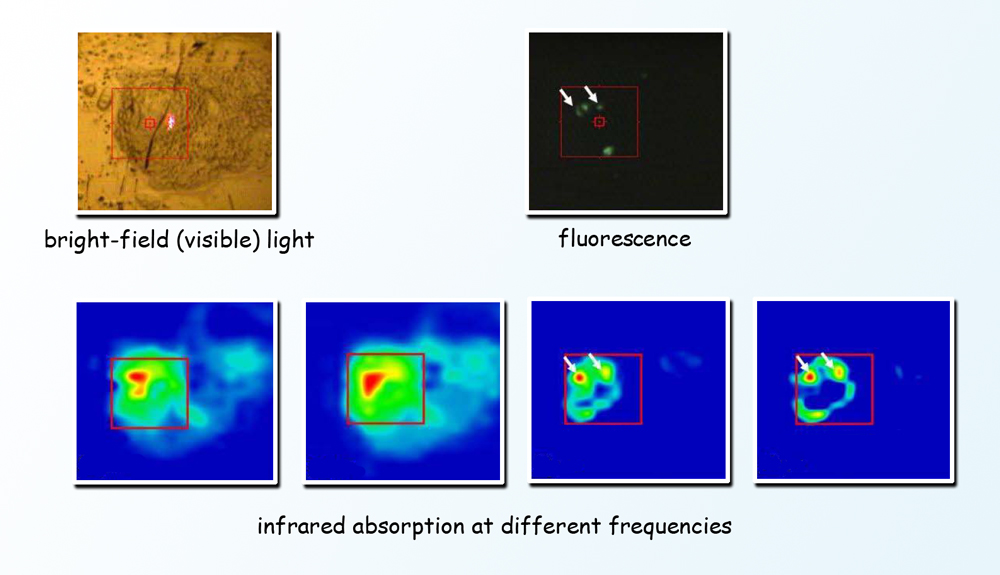
In the top panel, different modes of imaging of the same cell show the differences between visible light microscopy and fluorescence imaging, and in the lower panel, the images resulting from Fourier-transform infrared spectromicroscopy. Infrared absorption at different frequencies pinpoints different cell components at specific locations in the living cell. (Click on image for best resolution.)
A new technique takes off
“This experiment was a proof of the concept,” says Liang Chen. “We demonstrated the dynamics of protein phosphorylation in controlling differentiation in this biological system using synchrotron infrared spectromicroscopy, and we pointed the way to answering the many questions a biologist has to ask about measuring the coordination of specific processes in real time.”
Although in this first experiment the team was not able to follow individual cells continuously, they were able to monitor differentiation in groups of cultured PC12 cells in real time, without labeling or any other invasive procedure. It was the first step in an ambitious inquiry into the real-time biochemistry of living mammalian cells over the long term.
At beamline 1.4.3., with the help of new team members Kevin Loutherback and Rafael Gomez-Sjoberg, the team is designing equipment to maintain mammalian cells in a thin layer of culture media that will keep them healthy yet not interfere with the infrared beam, while automatically monitoring and adjusting temperature, humidity, and nutrient ratios, and removing waste products. This will allow data on individual cells to be gathered continuously throughout the entire phosphorylation process.
Meanwhile the Berkeley Synchrotron Infrared Structural Biology program at ALS beamline 5.4 is building multimodal facilities that will monitor cell development in human cells, bacteria, and plants, within soils, minerals, and other environments, via “hyperspectromicroscopy” – from the ultraviolet through visible light and deep into the infrared. Researchers will be able to choose the frequency window (or combination of windows) best suited to the sample and the conditions – in Holman’s words, “to watch almost everything at once.”
Says Holman, “Many researchers from the medical communities are interested in using the technology, and we are particularly interested in collaborating with university centers and private firms that are seeking a broad view of how promising drugs act within specific cells.”
Some of the projects will target Alzheimer’s disease, macular degeneration of the retina in diabetes, and mitochondrial diseases in children. In addition, specific processes like protein glycation can also be identified. Since different cells and different organisms respond differently, the eventual goal is to develop specific ways to screen the mechanisms of individual medicines.
###
“Synchrotron infrared measurements of protein phosphorylation in living single PC12 cells during neuronal differentiation,” by Chen et al, appears in Analytical Chemistry, online at http://pubs.acs.org/doi/full/10.1021/ac300308x.
The research was performed at the Advanced Light Source and the Berkeley Synchrotron Infrared Structural Biology Program, which are supported by the U.S. Department of Energy Office of Science.
Learn more about infrared beamlines at the Advanced Light Source at infrared.als.lbl.gov/.
The Advanced Light Source is a third-generation synchrotron light source producing light in the x-ray region of the spectrum that is a billion times brighter than the sun. A DOE national user facility, the ALS attracts scientists from around the world and supports its users in doing outstanding science in a safe environment. For more information visit www-als.lbl.gov/.
DOE’s Office of Science is the single largest supporter of basic research in the physical sciences in the United States, and is working to address some of the most pressing challenges of our time. For more information, please visit the Office of Science website at science.energy.gov/.
Lawrence Berkeley National Laboratory addresses the world’s most urgent scientific challenges by advancing sustainable energy, protecting human health, creating new materials, and revealing the origin and fate of the universe. Founded in 1931, Berkeley Lab’s scientific expertise has been recognized with 13 Nobel prizes. The University of California manages Berkeley Lab for the U.S. Department of Energy’s Office of Science. For more, visit www.lbl.gov.