Tag: photosynthesis
Chloro-phylling in the Answers to Big Questions

Showtime for Photosynthesis

A ‘Silver Bullet’ for the Chemical Conversion of Carbon Dioxide
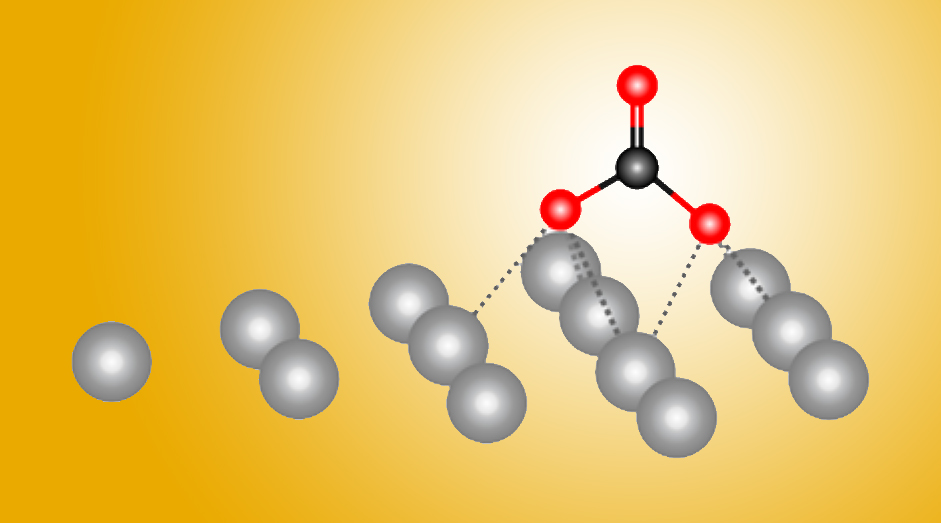
New Molecular Blueprint Advances Our Understanding of Photosynthesis
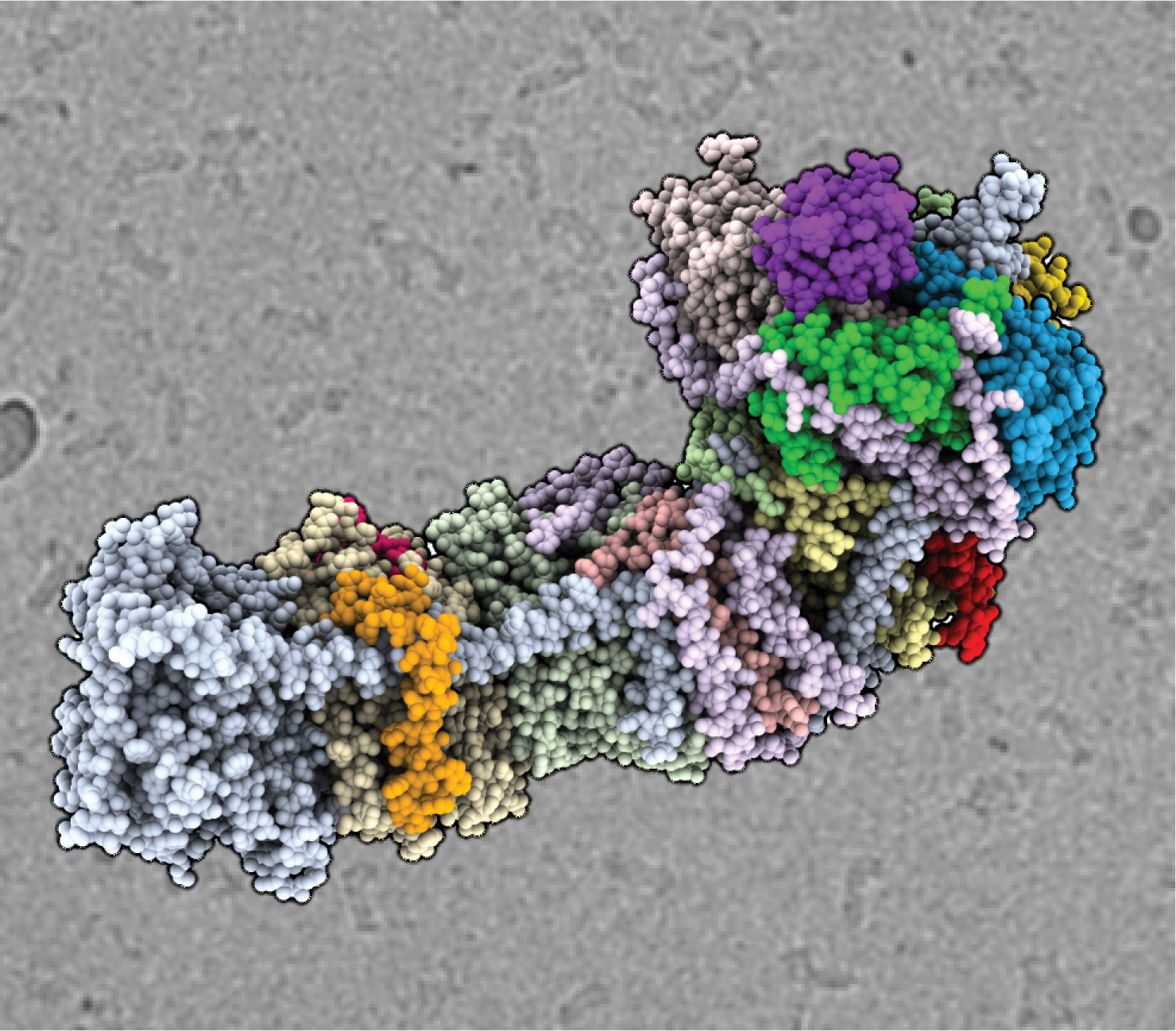
A Core−Shell Nanotube Array for Artificial Photosynthesis

Berkeley Lab Researchers ID Plant ‘Sunscreen’ Protein

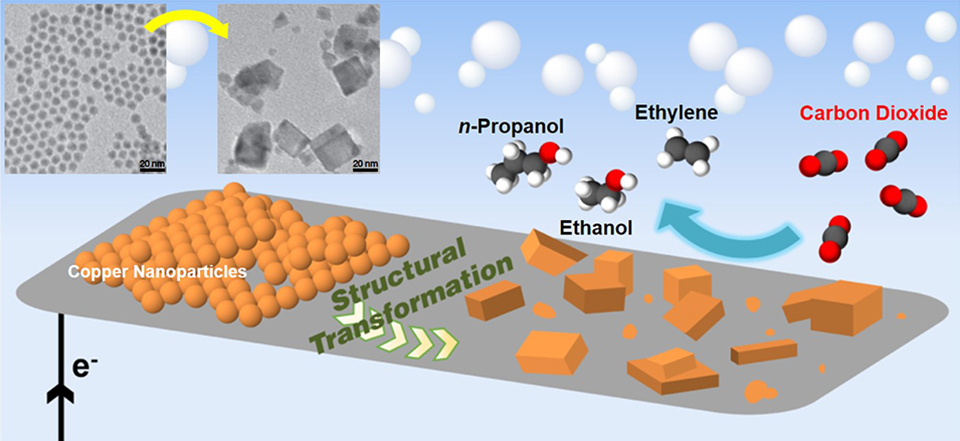
Copper Catalyst Yields High Efficiency CO2-to-Fuels Conversion
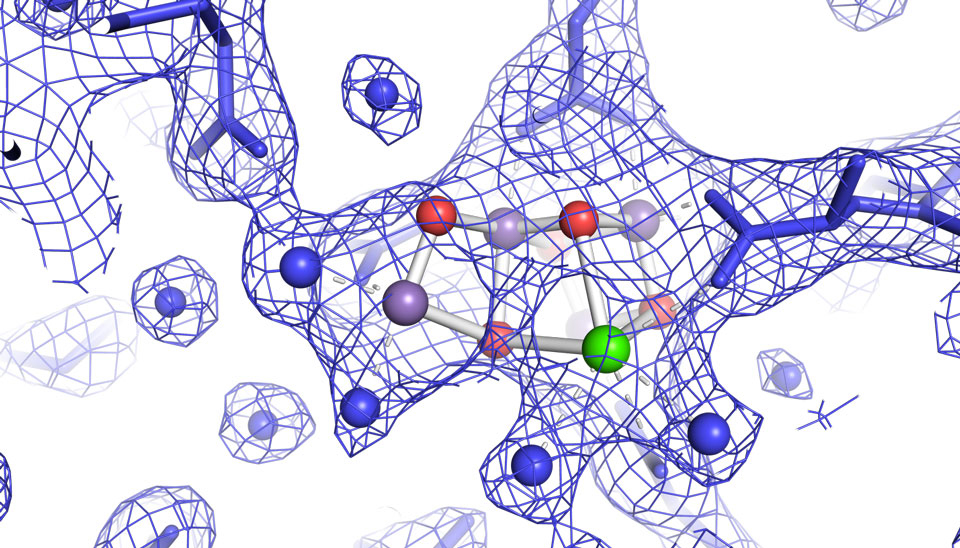
New Leaf Study Sheds Light on ‘Shady’ Past

X-Rays Capture Unprecedented Images of Photosynthesis in Action
Crop Yield Gets Boost with Modified Genes in Photosynthesis
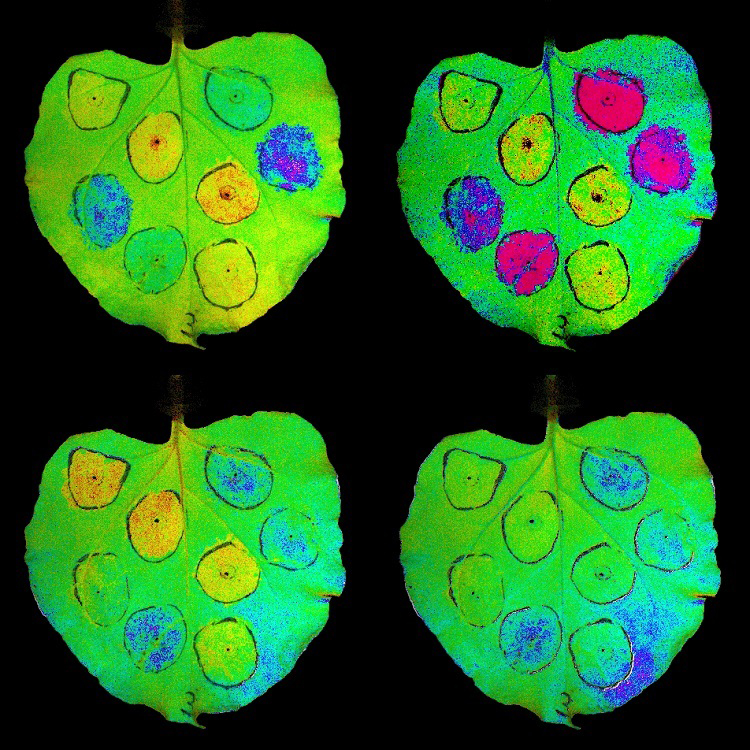
Solar Cells Get Boost with Integration of Water-Splitting Catalyst onto Semiconductor
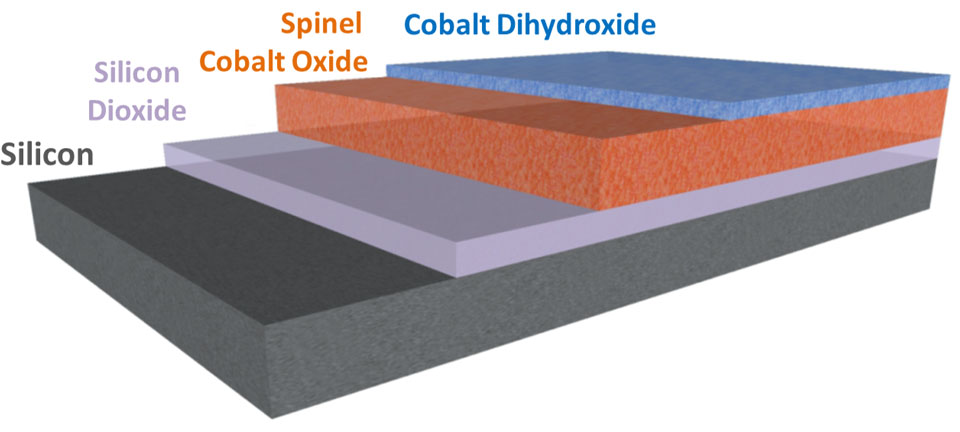
New Discovery Could Better Predict How Semiconductors Weather Abuse
