Adapted from a release by Robert Sanders at UC Berkeley
Vaccines save lives, as proven during the recent pandemic, but one component of most vaccines – including the Novavax COVID-19 vaccine – goes unheralded: a molecule or other compound that primes the immune system to mount a more robust defense against infection.
These so-called adjuvants are added in small quantities but have a big protective effect, particularly in infants with immature immune systems and older people with a declining immune response.
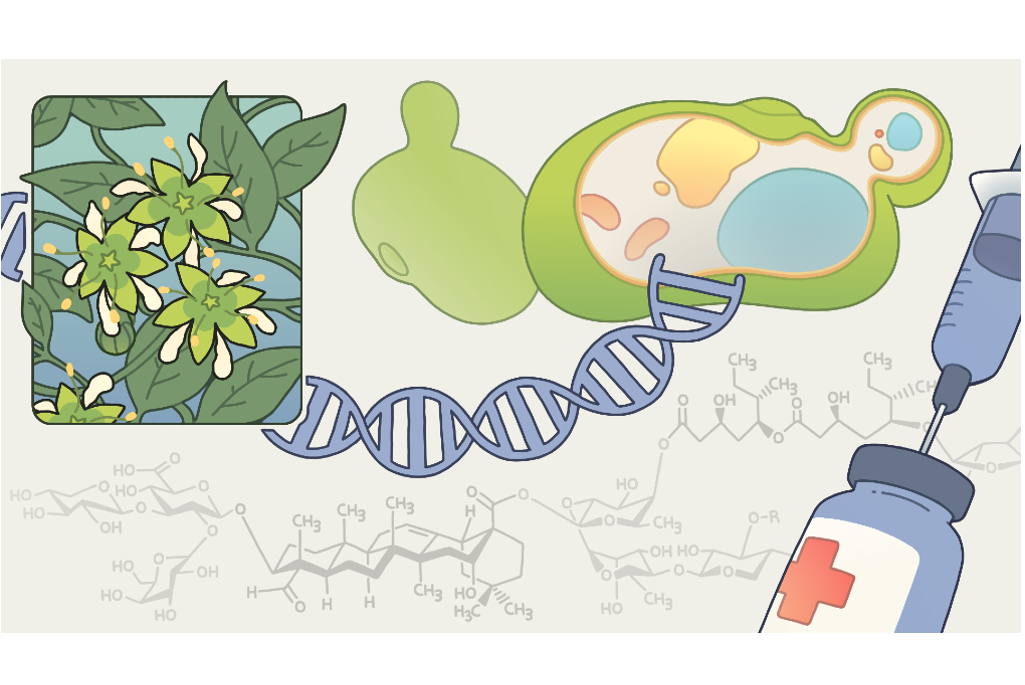
Yet, one of the strongest adjuvants, an extract of the Chilean soap bark plant, is so difficult to produce that it costs several hundred million dollars per kilogram (2.2 pounds).
UC Berkeley and Lawrence Berkeley National Laboratory (Berkeley Lab) scientists have now wielded the power of synthetic biology to produce the active ingredient of soap bark, a molecule called QS-21, in yeast. Producing compounds like this in yeast is not only cheaper, but more environmentally friendly, avoiding many of the caustic and toxic chemicals needed to extract the compound from plants.
While yields from the yeast-based process are still small – a few hundred dollars’ worth from a liter of broth – the feat promises to make one of the most effective adjuvants available more broadly and to lower the cost of vaccines, in general.
“During the pandemic, public health officers were really worried about QS-21 adjuvant availability because that only comes from one tree,” said Jay Keasling, UC Berkeley professor of chemical and biomolecular engineering and senior faculty scientist in Berkeley Lab’s Biosciences Area. “From a world health perspective, there’s a lot of need for an alternative source of this adjuvant.”
The production of QS-21 involved the insertion of 38 different genes from six organisms into yeast – building one of the longest biosynthetic pathways ever transplanted into any organism, Keasling said.
“The production of the potent vaccine adjuvant QS-21 in yeast highlights the power of synthetic biology to address both major environmental, as well as human, health challenges,” said former UC Berkeley postdoctoral fellow Yuzhong Liu, first author of the paper and now an assistant professor at Scripps Research in La Jolla, California.
The results were published today in the journal Nature.
Building upon malaria work
The benefit of adding an adjuvant to a vaccine was first noted in the 1920s, when alum – an aluminum salt – was discovered to boost the effectiveness of a diphtheria vaccine. Alum has since been added to many vaccines that use a portion of a pathogen – though not the infectious part – to induce immunity. Because adjuvants make vaccines more effective, they also allow doctors to use smaller doses of the active ingredient, called an antigen.
Not long after alum was discovered to boost the effectiveness of vaccines, a group of soap-like molecules was found to do the same. By the 1960s, researchers had focused on an extract of the Chilean soapbark tree (Quillaja saponaria) that strongly activates different components of the immune system to amplify the effect of giving a vaccine antigen alone. For the last 25 years, one component of that extract – QS-21 – has been one of the main non-aluminum adjuvants in vaccines, having been tested in more than 120 clinical trials. It is found in the shingles vaccine (Shingrix) given to older adults, a malaria vaccine (Mosquirix) currently used in children to protect against the parasite Plasmodium falciparum, and the Novavax’s SARS-COVID-19 vaccine.
QS-21 is produced today by stripping bark from the tree and chemically extracting and separating its many compounds, some of which are toxic. Though QS-21 is a complex molecule containing a terpenoid core and eight sugar molecules, it has been synthesized in the laboratory. But that synthesis takes 79 separate steps, starting from an intermediate chemical that itself has to be synthesized.
Keasling decided to try to recreate the synthesis process in yeast because for years he has successfully added genes to yeast to get them to make terpenes and other compounds with complex structures including vital medicines, biofuels, scents, and flavorings. In the early 2000s, he led the team that developed the first-ever biosynthetic pathway, producing a strain of yeast that makes artemisinin, a terpene-based antimalarial drug originally derived from a plant.
“This work builds on our malaria work,” he said. “We worked on the malaria therapy. Now, this could be an adjuvant for the malaria vaccines in the future.”
Adding the eight sugars proved challenging, as did balancing unsuspected interactions among enzymes in yeast. All this had to be accomplished without throwing off critical metabolic pathways that are needed for yeast growth.
“I mean, it makes the artemisinin biosynthetic pathway look like nothing,” Keasling said. “I am gratified that synthetic biology has come so far that we can now build a pathway to produce a molecule like QS-21. It’s a testament to how far the field has progressed in the last two decades.”
He and his lab colleagues, led by postdoctoral fellow Liu, worked closely with plant researcher Anne Osbourn at the John Innes Center in the United Kingdom. Osbourn had earlier teased out the many enzymatic steps involved in the soapbark tree’s production of natural QS-21. Over the past five years, as Osbourn discovered new steps in the process and tested them in tobacco plants, Keasling’s lab gradually added these new genes to yeast to replicate the synthetic steps.
“It was a great collaboration, because as soon as she’d get a new gene in the pathway, they’d send it our way, and we’d put it into yeast,” Keasling said. “It was also good for her, because she got a test of whether her tobacco assay was telling her the right thing.”
‘Everything from a single sugar’
Earlier this year, Osbourn and Keasling published the complete 20-step process by which the soapbark tree makes QS-21, reconstituted in tobacco. Unfortunately, tobacco is a test bed for plant chemistry, but not a scalable way to produce a chemical compound.
The new paper reconstitutes that process in yeast, with additional steps added because yeast do not contain some enzymes that naturally exist in plants. Currently, a liter of the fermenting bioengineered yeast can produce about 100 micrograms of QS-21 in three days, with a market value of about $200. But yeast biosynthesis is scalable.
“Even at the levels we’re producing it, it’s cheaper than producing it from the plant,” Keasling said.
The engineered yeast subsist only on sugar, which is an added advantage, he said.
“My whole thing is, I want to make everything from a single sugar. I just want to feed yeast glucose, because eventually we want this process to be scaled. And if you feed them a bunch of fancy intermediates, then it’s going to result in a process that is not scalable,” Keasling said. “In the end, I’d like to start with glucose, so when the production is performed in large tanks, they’re able to produce QS-21 as easily and inexpensively as possible.”
While Keasling plans to leave optimization of the process for large-scale production to others, he does hope to tweak the enzymatic steps he has introduced into yeast to produce variants of QS-21 that could potentially be more effective than QS-21. And yeast biosynthesis allows him to experiment with pruning the QS-21 molecule to see which portions can be eliminated without altering the molecule’s effectiveness. Additionally, the knowledge gained from this work will help scientists design efficient biomanufacturing processes for other valuable terpenes that are currently in limited supply.
With a similar goal to improve our public health system’s ability to respond to disease outbreaks, Berkeley Lab researchers are working on two projects under the Department of Energy’s Biopreparedness Research Virtual Environment (BRaVE) initiative: accelerating the development of new vaccines and antibody therapies for emerging pathogens using structural biology, and investigating using genetically modified bacteriophages to combat antibiotic resistant infections.
The research in this study was funded by an industrial grant.
###
Lawrence Berkeley National Laboratory (Berkeley Lab) is committed to delivering solutions for humankind through research in clean energy, a healthy planet, and discovery science. Founded in 1931 on the belief that the biggest problems are best addressed by teams, Berkeley Lab and its scientists have been recognized with 16 Nobel Prizes. Researchers from around the world rely on the Lab’s world-class scientific facilities for their own pioneering research. Berkeley Lab is a multiprogram national laboratory managed by the University of California for the U.S. Department of Energy’s Office of Science.
DOE’s Office of Science is the single largest supporter of basic research in the physical sciences in the United States, and is working to address some of the most pressing challenges of our time. For more information, please visit energy.gov/science.
It’s Hearty, It’s Meaty, It’s Mold

Making Renewable, Infinitely Recyclable Plastics Using Bacteria