Tag: chemical sciences
The Secret to Renewable Solar Fuels Is an Off-and-On Again Relationship

Berkeley Lab Researchers Receive DOE Early Career Research Awards

Berkeley Lab Researchers and Collaborators Elected into National Academy of Sciences

2 Berkeley Lab Scientists, Visiting Scientist Elected as New Members of Honorary Society

How JCAP Is Making Solar Fuels Shine

Scientists Discover New Clue Behind Age-Related Diseases and Food Spoilage

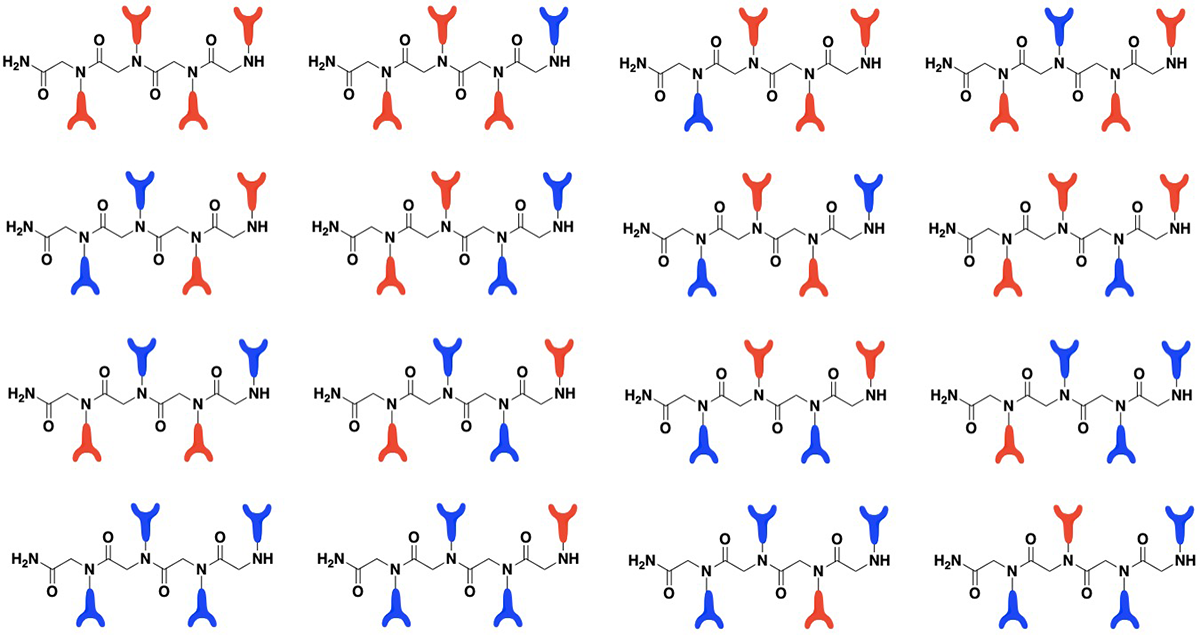
These Artificial Proteins Have a Firm Grasp on Heavy Metals
Six Berkeley Lab Scientists Named AAAS Fellows

Distinguished European Academy Elects Berkeley Lab Chemist Polly Arnold

A Single Dose for Good Measure: How an Anti-Nuclear-Contamination Pill Could Also Help MRI Patients
A Chemical Reaction Close-Up: New Technology Gives a Glimpse of Solar Fuel Generation in Action

The Chemistry of Art: Scientists Explore Aged Paint in Microscopic Detail to Inform Preservation Efforts
