Tag: chemical sciences
Berkeley Lab Appoints Polly Arnold as Chemical Sciences Division Director

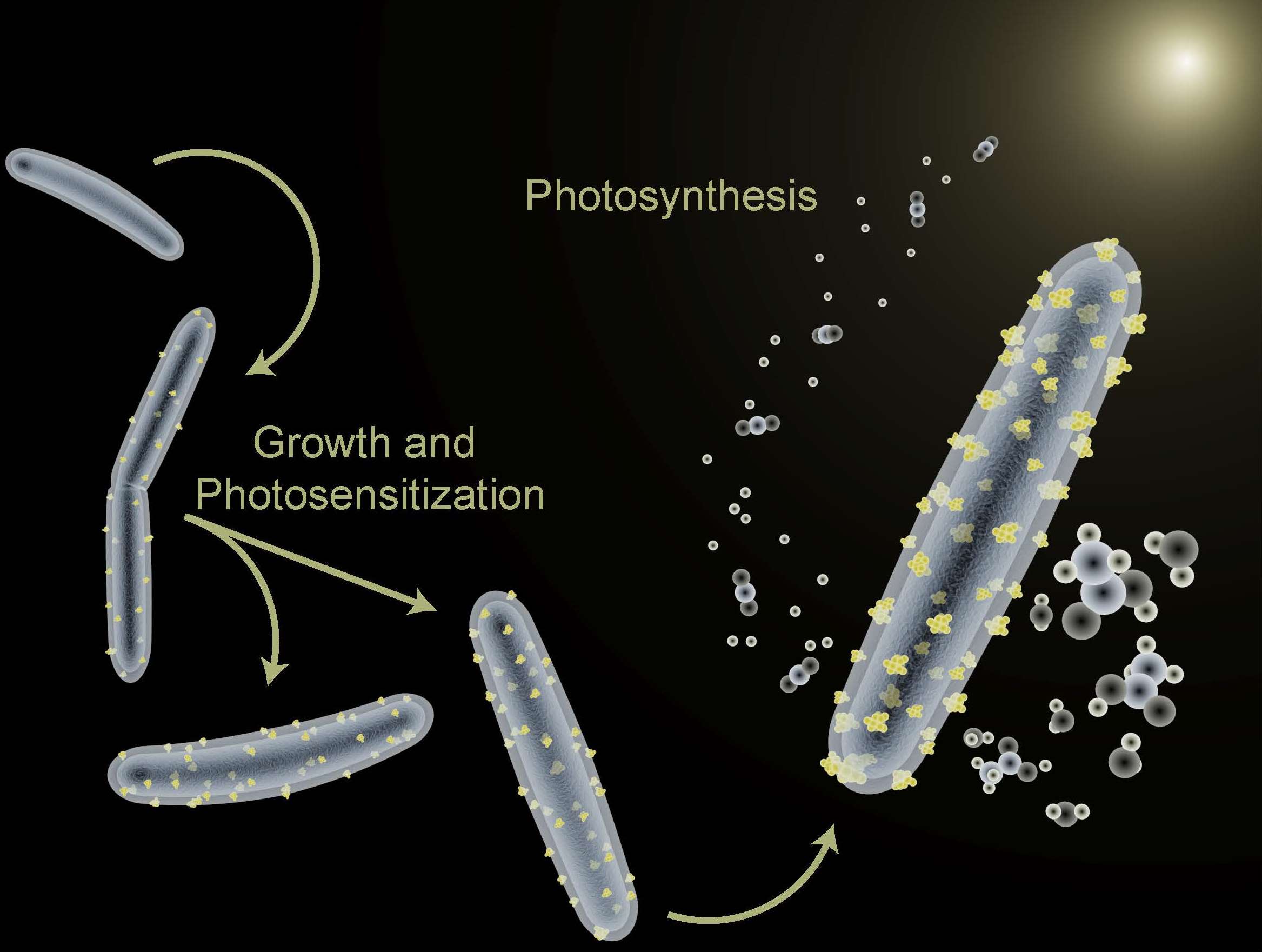
Here Comes the Sun: A New Framework for Artificial Photosynthesis

Separation Anxiety No More: A Faster Technique to Purify Elements

Royal Society of London Elects Two Berkeley Lab Scientists

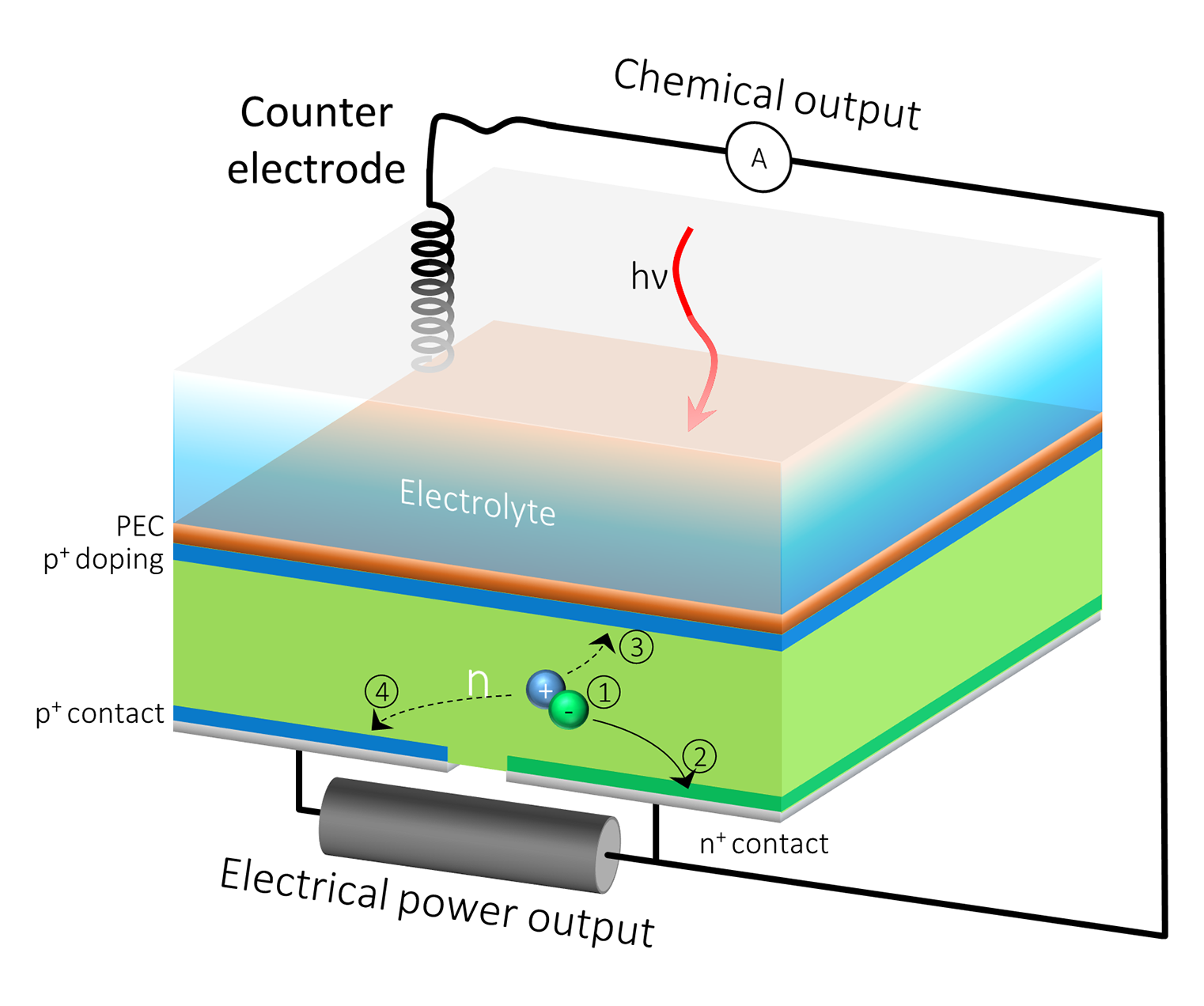
A Solar Cell That Does Double Duty for Renewable Energy
Scientists Present New Clues to Cut Through the Mystery of Titan’s Atmospheric Haze

Splitting Water: Nanoscale Imaging Yields Key Insights

Vote for Your Favorite Photos: 2018 Physics Photowalk Competition

4 Berkeley Lab-affiliated Scientists Elected as National Academy of Sciences Members

In Pursuit of Perfect Chemistry: A Vision for Unifying Catalysis
COSMIC Impact: Next-Gen X-Ray Microscopy Platform Now Operational

Chemical Sleuthing Unravels Possible Path to the Formation of Life’s Building Blocks in Space
