Tag: biology
Foundational AI Models to Accelerate Biological Discovery

Jay Keasling Receives Distinguished Scientist Fellow Award

A Detailed Look Inside Tsetse Flies
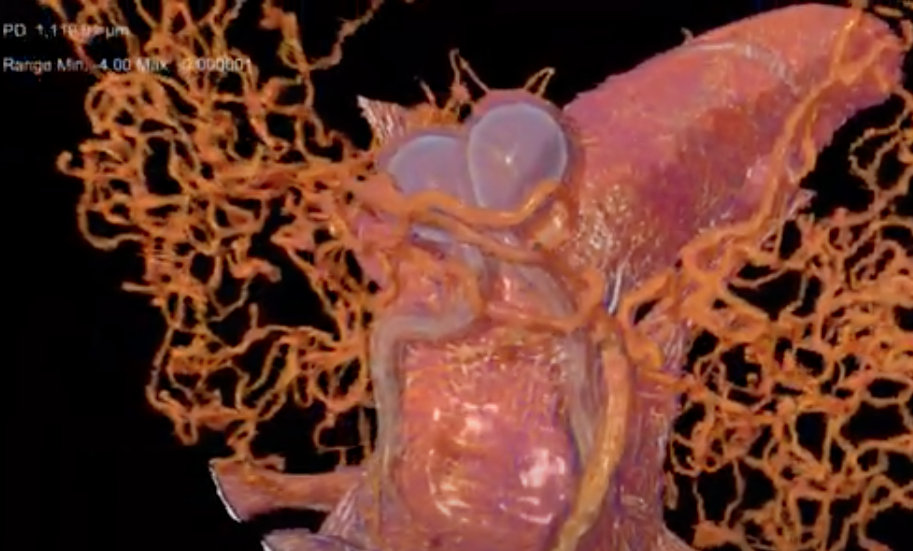
X-Rays Reveal Architectural Clues to the Crush-Resistance of Diabolical Ironclad Beetles

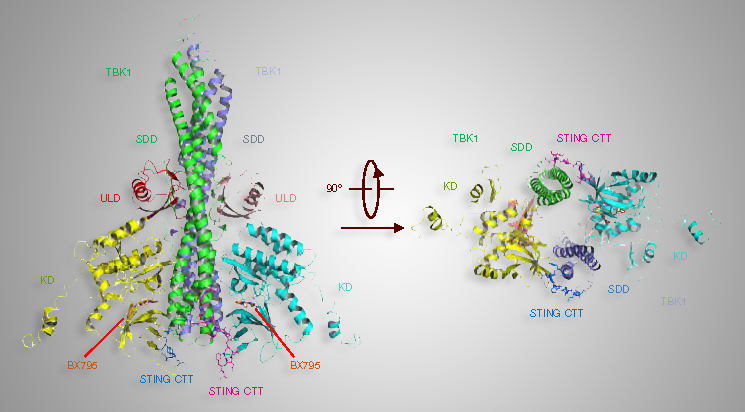
X-Ray Studies Key in Study Relating to Immune System-Signaling Protein
Photosynthesis Like a Moss

Infrared Beams Show Cell Types in a Different Light

Light-Emitting Nanoparticles Could Provide a Safer Way to Image Living Cells

Scientists Create Continuously Emitting Microlasers With Nanoparticle-Coated Beads

Diamond ‘Spin-Off’ Tech Could Lead to Low-Cost Medical Imaging and Drug Discovery Tools

Tau-tally Microtubular!

4 Berkeley Lab-affiliated Scientists Elected as National Academy of Sciences Members
